2024
77.Partial Thermal Condensation Mediated Synthesis of High‐Density Nickel Single Atom Sites on Carbon Nitride for Selective Photooxidation of Methane into Methanol
Pawan Kumar, Peter Antal, Xiyang Wang, Jiu Wang, Dhwanil Trivedi, Ondřej František Fellner, Yimin A. Wu, Ivan Nemec, Vinicius Tadeu Santana, Josef Kopp, Petr Neugebauer, Jinguang Hu, Md Golam Kibria, Subodh Kumar, Small, 2024, accepted
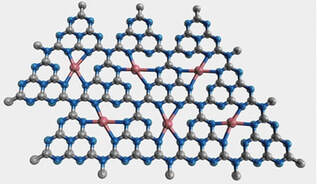
Direct selective transformation of greenhouse methane (CH4) to liquid oxygenates (methanol) can substitute energy-intensive two-step (reforming/Fischer–Tropsch) synthesis while creating environmental benefits. The development of inexpensive, selective, and robust catalysts that enable room temperature conversion will decide the future of this technology. Single-atom catalysts (SACs) with isolated active centers embedded in support have displayed significant promises in catalysis to drive challenging reactions. Herein, high-density Ni single atoms are developed and stabilized on carbon nitride (NiCN) via thermal condensation of preorganized Ni-coordinated melem units. The physicochemical characterization of NiCN with various analytical techniques including HAADF-STEM and X-ray absorption fine structure (XAFS) validate the successful formation of Ni single atoms coordinated to the heptazine-constituted CN network. The presence of uniform catalytic sites improved visible absorption and carrier separation in densely populated NiCN SAC resulting in 100% selective photoconversion of (CH4) to methanol using H2O2 as an oxidant. The superior catalytic activity can be attributed to the generation of high oxidation (NiIII═O) sites and selective C─H bond cleavage to generate •CH3 radicals on Ni centers, which can combine with •OH radicals to generate CH3OH.
2023
76.Honey-like Odor Meets Single-Ion Magnet: Synthesis, Crystal Structure, and Magnetism of Cobalt(II) Complex with Aromatic Trans-Cinnamic Acid
Petr Halaš, Ivan Nemec and Radovan Herchel, Magnetochemistry, 2023, 9(11), 229; https://doi.org/10.3390/magnetochemistry9110229
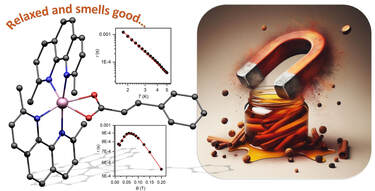
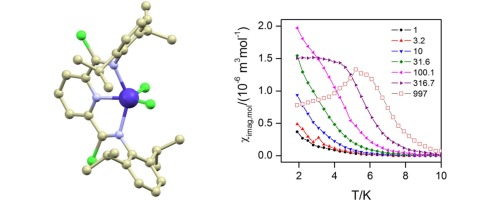
The hexacoordinate Co(II) complex [Co(neo)2(cin)][BPh4]·½Me2CO (1·½Me2CO) containing trans-cinnamic acid (Hcin) and neocuproine (neo) was prepared. The compound 1·½Me2CO was characterized via single-crystal X-ray analysis, FT-IR spectroscopy, and magnetic measurements. The coordination polyhedron of the complex cation adopts a deformed octahedron shape, and cinnamate exhibits a bidentate mode of coordination, which is unusual for mononuclear Co(II) cinnamate complexes. The analysis of DC magnetic measurements with zero-field splitting (ZFS) spin Hamiltonian revealed large magnetic anisotropy defined by the axial ZFS parameter D = +53.2 cm−1. AC susceptibility measurements revealed the slow relaxation of magnetization under the applied field; thus, 1·½Me2CO behaves as a field-induced single-molecule magnet. The analysis of magnetic properties was also supported by CASSCF/NEVPT2 calculations.
75. 2-Formylphenoxyacetic acid Schiff bases: a promising ligand scaffold for readily available trigonal prismatic Co(ii) single-ion magnets
Kamil Kotrle, Ivan Nemec, Peter Antal, Kamila Petrželová, Erik Čižmár and Radovan Herchel, Inorg. Chem. Front., 2023, 10, 7319-7332
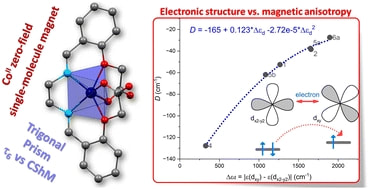
This article presents a series of six mononuclear Co(II) complexes 1–6 featuring ligands derived from a hexadentate Schiff base family, originating from the condensation of (2-formylphenoxy)acetic acid with various diamines. Notably, these complexes uniquely prefer a trigonal prism geometry, presenting a novel approach to synthesizing complexes with this distinctive shape. The compounds were characterized by elemental analysis, FT-IR spectroscopy, and single-crystal and powder XRD techniques. Furthermore, the magnetism was investigated by DC and AC magnetic measurements and also complemented by X-band EPR spectroscopy. The results reveal that the prepared complexes behave as field-induced single-molecule magnets, characterized by a substantial negative axial zero-field splitting D-parameter and spin reversal energetic barrier Ueff reaching values up to 72 K. The theoretical methods based on CASSCF/NEVPT2 calculations were applied to rationalize their magnetic properties. Moreover, these complexes hold promising potential for further functionalization, offering opportunities to enhance their properties, particularly towards developing zero-field single-molecule magnets as evidenced by the slow relaxation of magnetization in zero static magnetic field observed for the zinc-diluted complex 1Zn.
74. Tetracoordinate Co(II) Complexes with Semi-Coordination as Stable Single-Ion Magnets for Deposition on Graphene
Jorge Navarro-Giraldo, Jakub Hrubý, Šárka Vavrečková, Ondřej František Fellner, Lubomír Havlíček, DaVonne Henry, Shehan de Silva, Radovan Herchel, Miroslav Bartos, Ivan Salitros, Vinicius Tadeu Santana, Paola Barbara, Ivan Nemec and Petr Neugebauer, Phys. Chem. Chem. Phys., 2023, 25, 29516-29530
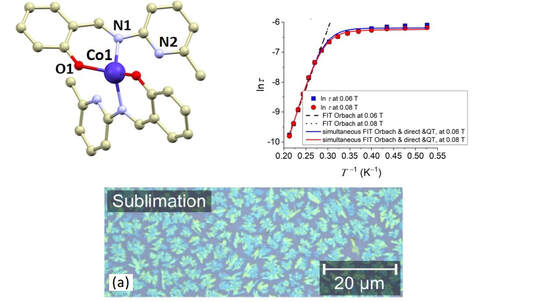
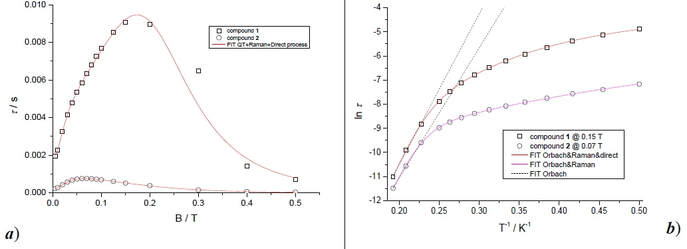
We present a theoretical and experimental study of two tetracoordinate Co(II)-based complexes with semi-coordination interactions, i.e., non-covalent interactions involving the central atom. We argue that such interactions enhance the thermal and structural stability of the compounds, making them appropriate for deposition on substrates, as demonstrated by their successful deposition on graphene. DC magnetometry and high-frequency electron spin resonance (HF-ESR) experiments revealed an axial magnetic anisotropy and weak intermolecular antiferromagnetic coupling in both compounds, supported by theoretical predictions from complete active space self-consistent field calculations complemented by N-electron valence state second-order perturbation theory (CASSCF-NEVPT2), and broken-symmetry density functional theory (BS-DFT). AC magnetometry demonstrated that the compounds are field-induced single-ion magnets (SIMs) at applied static magnetic fields, with slow relaxation of magnetization governed by a combination of quantum tunneling, Orbach, and direct relaxation mechanisms. The structural stability at ambient conditions and after deposition was confirmed by X-ray photoelectron spectroscopy (XPS) and Raman spectroscopy. Theoretical modeling by DFT of different configurations of these systems on graphene revealed n-type doping of graphene originating from electron transfer from the deposited molecules, confirmed by electrical transport measurements and Raman spectroscopy.
73. Rh(III) and Ru(II) complexes with phosphanyl–alkylamines: inhibition of DNA synthesis induced by anticancer Rh complex
Alena Mrkvicová, Eva Peterová, Ivan Nemec, Radka Křikavová, Darina Muthná, Radim Havelek, Petra Kazimírová, Martina Řezáčová, Pavel Štarha, Future Med. Chem. 2023, 15, 17, 1583–1602
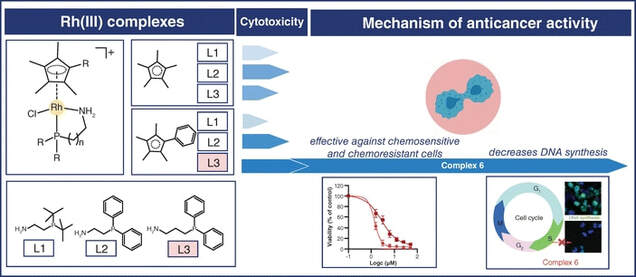
Nine rhodium(III)and ruthenium(II) complexes were developed and screened for their anticancer activity on a panel of human cancer cell lines. The best-performing rhodium(III) complex (6) showed high activity in ovarian cancer cells, including the variant resistant to the conventional anticancer drug cisplatin, while it was less effective towards non-cancerous lung fibroblasts. In cancer cells, compound 6 induced a modification of the cell cycle connected with a significant decrease in DNA synthesis, which was not observed for cisplatin. The effect of 6 on the expression of proteins related to the cell cycle modification was analysed by quantitative PCR and western blot in cancer cells and the results indicated a p21-independent mode of anticancer action, which is different from cisplatin.
72.Impact of central atom and halido ligand on structure, antiproliferative activity and selectivity of half-sandwich Ru(II) and Ir(III) complexes with a 1,3,4-thiadiazole-based ligand.
Radka Křikavová, Michaela Romanovová, Zuzana Jendželovská, Martin Majerník, Lukáš Masaryk, Pavel Zoufalý, David Milde, Jan Moncol, Radovan Herchel, Rastislav Jedželovský and Ivan Nemec, Dalton Trans. 2023, 52, 12717-12732
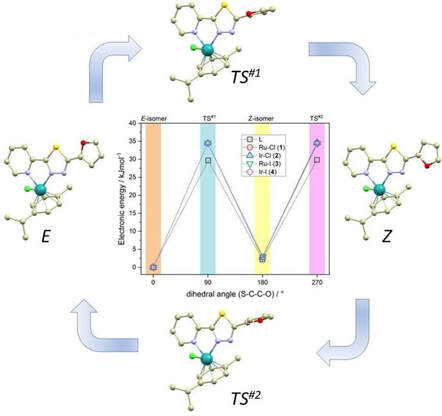
Half-sandwich complexes [Ru(η6-pcym)(L1)X]PF6 (1, 3), [Ir(η5-Cp*)(L1)X]PF6 (2, 4) featuring a thiadiazole-based ligand L1 (2 (furan‐2‐yl)‐5‐(pyridin‐2‐yl)‐1,3,4‐thiadiazole) were synthesized and characterized, including single-crystal X-ray diffraction (X = Cl or I, pcym = p-cymene, Cp* = pentamethylcyclopentadienyl). The structures of the molecules were analysed and interpreted using computational methods such as Density Functional Theory (DFT) and Quantum Theory of Atoms in Molecules (QT-AIM). 1H NMR spectroscopic study showed that complexes 1-3 exhibited hydrolytic stability while 4 underwent partial iodido/chlorido ligand exchange in phosphate-buffered saline. Moreover, 1-4 demonstrated the ability to oxidize NADH (reduced nicotinamide adenine dinucleotide) to NAD+ with Ir(III) complexes 2 and 4 displaying higher catalytic activity compared to their Ru(II) analogues. None of the complexes interacted with reduced gluthathione (GSH). Additionally, 1-4 exhibited greater lipophilicity than cisplatin. The in vitro biological analyses were performed in healthy cell lines (CCD-18Co colon and CCD-1072Sk foreskin fibroblasts) as well as in cisplatin-sensitive (A2780) and -resistant (A2780cis) ovarian cancer cell lines. The results indicated that Ir(III) complexes 2 and 4 had no effect on human fibroblasts, demonstrating their selectivity. In contrast, complexes 1 and 4 exhibited moderate inhibitory effects on the metabolic and proliferation activity of the cancer cells tested (selectivity index SI > 3.4 for 4 and 2.6 for cisplatin; SI = IC50(A2780)/IC50(CCD-18Co)), including the cisplatin-resistant cancer cell line. Based on these findings, it is possible to emphasize that mainly complex 4 could represent a further step in the development of selective and highly effective anticancer agents, particularly against resistant tumour types.
71. Mn(III)–Salen Complexes with Metallophilic Interactions
Tomáš Šilha, Radovan Herchel and Ivan Nemec, Crystals 2023, 13(8), 1217
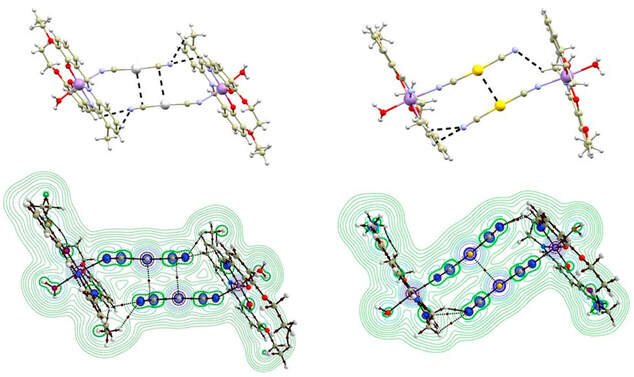
We synthesized a series of five novel Mn–salen-based compounds (1a–1c, 2a, 2b) through the reaction between precursor chloride complexes and potassium silver/gold dicyanide. The prepared compounds were structurally and magnetically characterized. Our findings revealed that all the Mn(III) central atoms exhibited an axially elongated coordination polyhedron, leading to the observation of axial magnetic anisotropy as indicated by the negative axial magnetic parameter D, which was determined through fitting the experimental magnetic data and supported by theoretical CASSCF/NEVPT2 calculations. Furthermore, we observed magnetic-exchange interactions only in compounds with a special supramolecular topology involving O–H···O hydrogen-bonded dimers. In these cases, the weak magnetic exchange (J/cm−1 = −0.58(2) in 1b and −0.73(7) in 2b) was mediated by the O–H···O hydrogen bonds. These findings were further supported by BS–DFT calculations, which predicted weak antiferromagnetic exchanges in these complexes and ruled out exchange interactions mediated by diamagnetic cyanido metallo–complex bridges. Additionally, we investigated the observed Ag···π (1b) and Au···Au (2b) interactions using QT–AIM calculations, confirming their non-covalent nature. We compared these results with previously reported Mn–salen-based compounds with metallophilic interactions arising from the presence of the [Ag/Au(CN)2]− bridging units.
70.Neutral Cobalt(II)-Bis(Benzimidazole)Pyridine Field-Induced Single-Ion Magnets for Surface Deposition
Jana Juráková, Ondřej František Fellner, Sören Schlittenhardt, Šárka Vavrečková, Ivan Nemec, Radovan Herchel, Erik Cizmar, Vinicius Tadeu Santana, Milan Orlita, Denis Gentili, Giampiero Ruani, Massimiliano Cavallini, Petr Neugebauer, Mario Ruben and Ivan Salitros, Inorg. Chem. Front. 2023, 10, 5406-5419
Two novel hexacoordinate Co(II)-based single-ion magnets were prepared and characterised. Both neutral complexes feature the metal centre coordinated with one terminal and one bidentate nitrate anions along with tridentate derivatives of 2,6-bis(1H-benzimidazole-2-yl)pyridine ligand containing either n-octyl (complex 1) or n-dodecyl (complex 2) chains. The presence of long aliphatic chains ensures the solubility in the low polar and volatile solvents frequently used for lithography patterning. This enabled the preparation of microstructural layers and patterns on technologically relevant substrates by easy-to-handle and low-cost wet lithographic techniques. On the other hand, attempts for surface deposition via sublimation were not successful due to thermal instability. The electronic structure of complexes typically features an orbitally non-degenerate ground state well-separated from the lowest excited state, which allows to analyse the magnetic anisotropy by spin Hamiltonian approach. Zero-field splitting parameters obtained from CASSCF-NEVPT2 calculations and from analysis of magnetic data suggest that both compounds display positive axial D parameters within a range of 17-25 cm-1. Combined results from high-field electron paramagnetic resonance (X-band and HF-EPR) and Fourier-Transform infrared magnetic spectroscopy (FIRMS) simulated with the spin Hamiltonian provided the axial and rhombic zero-field splitting terms D = +23.7 cm-1 for complex 1 and D = +24.2 cm-1 for complex 2, together with pronounced rhombicity in the range E/D ≈ 0.15-0.19 for both compounds. Dynamic magnetic investigations have revealed the field-induced slow relaxation of magnetisation, with maximal relaxation times (τ) of 7.6 ms for 1 and 0.8 ms for 2. This relaxation is governed via a combination of several relaxation mechanisms, among which the quantum tunnelling was efficiently suppressed by the applied static magnetic field. The effective barrier of spin reversal Ueff = 77(4) K for 1 and Ueff = 70(2) K for 2 are consistent with the expected values calculated using the ZFS parameters.
69.Axially Chiral Sulfonic Acids for Brønsted Acid Catalysis: 8-Benzoimidazolylnaphthalene-1-sulfonic Acids and Their Derivatives
Monika Tomanová, Iva Vaňková, Daniel Toman, Adam Přibylka, Ivan Nemec, and Petr Cankař, The Journal Of Organic Chemistry, 2023, 88, 13, 9265–9276
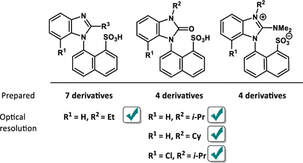
A new type of axially chiral sulfonic acid was developed. The synthesis is based on cheap commercially available materials and a practical method for optical resolution via diastereomeric salt formation, which can provide both enantiomers. Eleven benzoimidazolylnaphthalenesulfonic acids were prepared and four of them were isolated as pure and stable atropisomers. Moreover, several of these sulfonic acids were transformed into triflyl imides to further expand the range of dissociation constants.
68. Anti-myeloma pro-apoptotic Pt(II) diiodido complexes
Lukáš Masaryk, Denisa Weiser Drozdkova, Karolina Słoczyńska, Jan Moncol, David Milde, Radka Křikavová, Justyna Popiół, Elsbieta Pekala, Katarina Ondruskova, Ivan Nemec, Katerina Smesny Trtkova, Pavel Starha, Inorg. Chem. Front. 2023, 10, 3307-3318
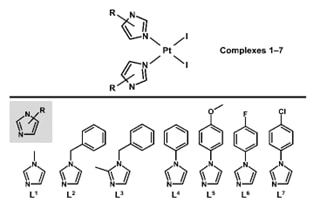
Platinum-based agents unwaveringly hold their prominent position in cancer treatment. Current research emphasizes finding novel complexes for hard-to-treat cancers with minimum side effects, capable of overcoming resistance. This work presents easy-to-prepare diiodidoplatinum(II) complexes cis-[PtI2(Ln)2] (1–7) with imidazole derivatives (Ln), which were considerably effective against multiple myeloma cell lines U266B1 and KMS12-PE. The leading compound 6 (at 3 μM concentration) extraordinarily reduced viability of U266B1 and KMS12-PE myeloma cells to 3.0% and 1.1%, respectively, and exceeded the conventional platinum-based anticancer drug cisplatin (93.1% and 88.3%, respectively) that is used clinically for the combination therapy of multiple myeloma. Complex 6 was significantly more effective in inducing apoptosis in KMS12-PE cells without interleukin-6 (IL-6) expression than in U266B1 cells with IL-6 expression. Complex 6 also induced apoptosis in co-culture of KMS12-PE with non-cancerous stromal fibroblasts (HS-5), and displayed markedly lower activity in the HS-5 stromal fibroblast cells than in myeloma cells, pointing out its pharmocologically prospective selectivity towards the cancer cells over the normal ones. No caspase 3/7 activity was detected in apoptotic KMS12-PE cells treated by complex 6 indicating a different mechanism of apoptosis action from cisplatin. This work demonstrates that simple non-classical platinum(II) complexes represent a new perspective for a monotherapy of hard-to-treat multiple myeloma.
67. Variation of Spin-Transition Temperature in the Iron (III) Complex Induced by Different Compositions of the Crystallization Solvent
Ivan Nemec, Lucie Kotaskova, Radovan Herchel, Crystal, Growth&Design, 2023, 23, 3, 1323–1329 Citations
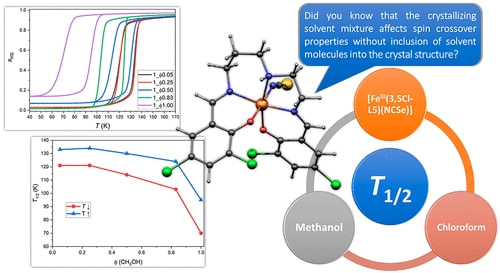
We crystallized the Schiff-base iron(III) spin-crossover complex [Fe(3,5Cl-L5)(NCSe)] from different two-component solvent mixtures containing methanol and chloroform (Φ = V(CH3OH)/V(solvent) = 0.05, 0.25, 0.50, 0.83, and 1.00). The obtained crystalline products were characterized by X-ray diffraction, and it was confirmed that they are all composed of the same crystalline phase, and they do not contain any crystal solvent. However, significant differences in magnetic properties were observed, and thermal hysteresis changed from (in K) 121T↓ and 134T↑ for Φ = 0.05 and 0.25, down to 72T↓ and 96T↑ for Φ = 1.00. The crystal structures of the low-spin and high-spin phases were studied theoretically and experimentally.
66. Antibacterial study on nickel and copper dicarboxylate complexes
Ivana Loubalová, EvaZahradníková, LukášMasaryk, IvanNemec, Lucie Hochvaldová, Aleš Panáček, Libor Kvítek, Renata Večeřová, Marcin Świątkowski, Pavel Kopel, Inorg. Chim. Acta, Volume 545, 2023, 121273, Citations
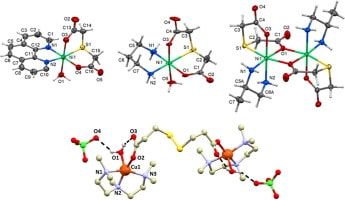
Series of nickel and copper complexes with dicarboxylic acids (2,2′-thiodiacetic acid, 3,3′–thiodipropionic acid, 3,3′-dithiodipropionic acid, and fumaric acid) and N-donor ligands (1,2–diaminopropane, 1,3-diaminopropane, 1,10-phenanthroline and N,N,N′,N″,N″–pentamethyldiethylenetriamine) were synthesized and characterized using elemental analysis, IR spectroscopy and single-crystal X-ray diffraction. These coordination compounds were tested for their antibacterial activities in vitro. The complexes demonstrated moderated antibacterial activity against Gram-positive and Gram-negative bacteria and could potentially be applied to treat skin wounds.
2022
65. New Ferrocene-Based Metalloligand with Two Triazole Carboxamide Pendant Arms and Its Iron(II) Complex: Synthesis, Crystal Structure, 57Fe Mössbauer Spectroscopy, Magnetic Properties and Theoretical Calculations
Peter Antal, Ivan Nemec, Jiří Pechoušek and Radovan Herchel, Inorganics, 2022, 10(11), 199, Citations
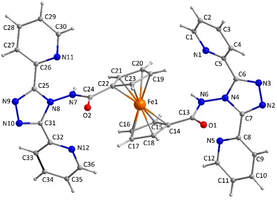
The new ferrocene-based metalloligand bis (N-4-[3,5-di-(2-pyridyl)-1,2,4-triazoyl])ferrocene carboxamide (L) was prepared through derivatization of 1,1′-ferrocenedicarboxylic acid with 4-amino-3,5-di(pyridyl)-4H-1,2,4-triazole. The composition and purity of L in the solid state was determined with elemental analysis, FT-IR spectroscopy, and its crystal structure with single-crystal X-ray analysis, which revealed that the substituted cyclopentadienyl rings adopt the antiperiplanar conformation and the crystal structure of L is stabilized by O–H···N and N–H···O hydrogen bonds. The molecular properties of L in solution were investigated with NMR and UV-VIS spectroscopies, and cyclic voltammetry disclosed irreversible redox behavior providing one oxidation peak at E1/2 = 1.133 V vs. SHE. Furthermore, the polymeric FeII complex {Fe(L)(C(CN)3)2}n (1) was prepared and characterized with elemental analysis, FT-IR spectroscopy, 57Fe Mössbauer spectroscopy, and magnetic measurements. The last two methods confirmed that a mixture of low- and high-spin species is present in 1; however, the spin crossover properties were absent. The presented study was also supported by theoretical calculations at the DFT/TD-DFT level of theory using TPSS and TPSSh functionals.
64. p-TSA-Mediated Four-Component Reaction: One-Step Access to Mesoionic 1H-Imidazol-3-ium-4-olates, Direct NHC Precursors
Naděžda Cankařová, Ivan Nemec, Viktor Krchnak, Advanced Synthesis & Catalysis, 2022, 364 (17), 2996-3003, Citations
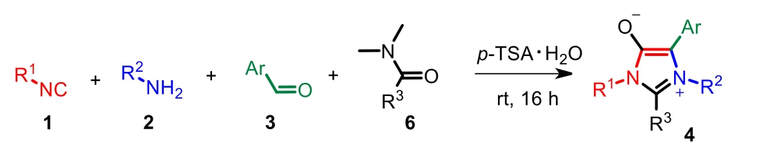
p-TSA-mediated four-component condensation is described. The reaction of an isocyanide, amine, aldehyde, and amide catalyzed by p-TSA afforded diversely substituted mesoionic 1H-imidazol-3-ium-4-olates in one step. These products were obtained in good to excellent HPLCpurity under mildreaction conditions. This formation of mesoionic compounds, structurally related to sydnones and münchnones, represent attractive intermediates and direct precursors for anionicN-heterocyclic carbenes. Atom economy, time efficiency, and commercially available starting materials make this one-pot synthesis user-friendly.
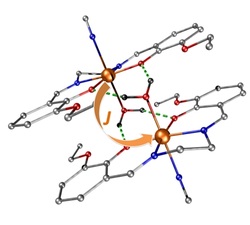
63. Weak antiferromagnetic interaction in Cu(II) complex with semi-coordination exchange pathway
Ľubomír Havlíček, Radovan Herchel, Ivan Nemec, Petr Neugebauer, Polyhedron, 2022, Volume 223, 115962, Citations
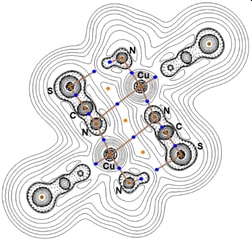
We present structural and magnetic studies of complex [Cu(dpt)(NCS)2], 1a, dpt = bis(3-aminopropyl)amine. A
weak antiferromagnetic interaction mediated by a Cu⋯π non-covalent interaction was identified by magnetometry
measurements, and the exchange magnitude (J = -2.24 cm−1, J stands for isotropic magnetic exchange constant)
was determined. These results were supported by theoretical calculations (DFT) and the nature of the Cu⋯
π interaction was studied by QT-AIM. It was confirmed that the Cu⋯ π non-covalent interaction could be described
as semi-coordination. The obtained results were compared to those previously reported for the isomorphous
complex [Co(dpt)(NCS)2].
62. Anticancer half-sandwich Ir(III) complex and its interaction with various biomolecules and their mixtures - a case study with ascorbic acid
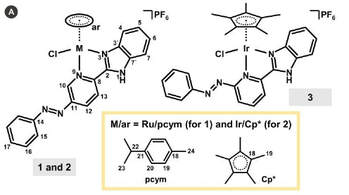
Lukáš Masaryk, Jakub Orvoš, Karolina Słoczyńska, Radovan Herchel, Jan Moncol, David Milde, Petr Halaš, Radka Krikavova, Paulina Koczurkiewicz, Elsbieta Pekala, Róbert Fischer, Ivan Salitros, Ivan Nemec and Pavel Starha, Inorg. Chem. Front., 2022, 9, 3758 - 3770, Citations
The extent of interactions with various biomolecules is a crucial feature of newly developed metallodrugs, worthy of
thorough investigation, as its understanding helps uncover the fate of these xenobiotics in the physiological environment.
In this work, promisingly cytotoxic half-sandwich complexes [Ru(η6-pcym)Cl(L1)]PF6 (1), [Ir(η5-Cp*)Cl(L1)]PF6 (2) and [Ir(η5-
Cp*)Cl(L2)]PF6 (3), with 2-{n-[(E)-phenyldiazenyl]pyridin-2-yl}-1H-benzimidazole as a bidentate N-donor azo ligand (n = 5 for
L1 and 6 for L2 ; pcym = p-cymene, Cp* = pentamethylcyclopentadienyl), were subjected to an extensive and detailed study
of interactions with a plethora of extra- and intracellular biologically relevant molecules. For the first time in the field of
anticancer half-sandwich complexes, the mixtures of 3 with ascorbic acid (ASA) and its combinations with reduced
glutathione (GSH) and/or reduced nicotinamide adenine dinucleotide (NADH) were studied. Complex 3 undergoes the azo
bond reduction when mixed with NADH or ASA, which oxidize to NAD+ and dehydroascorbate (DHA), respectively.
Intriguingly, the presence of the natural antioxidant ASA has a relevant prooxidative impact towards GSH, which is connected
with the ASA recovery from DHA. Although the azo bond of L2 involved in 3 seems to be the reaction centre for the
dehydrogenation reactions of the biomolecules, L2 by itself is a negligible oxidant and thus complexation in 3 represents a
necessary prerequisite for the redox reactions.
61. Solvent-directed morphological transformation in covalent organic polymers
Subodh Kumar, Xuan Thang Cao, Ivan Nemec, Rajender Singh Varma, Frontiers in Materials, 2022, 9, Citations
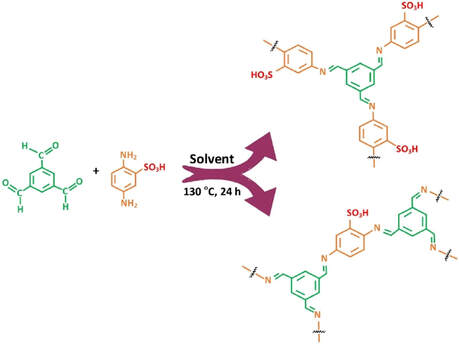
Synthesis of bi-functional covalent organic polymers in two distinctive morphologies has Q8
been accomplished by simply switching the solvent from DMF to DMSO when 1,3,5-
tribenzenecarboxyldehyde and 2,5-diaminobenzene sulfonic acid were reacted via Schiff
base condensation reaction to afford covalent organic polymers (COPs) encompassing
flower (F-COPDMF)- and circular (C-COPDMSO)-type morphologies. Chemical and
morphological natures of the synthesized COPs were compared by characterization
using TEM, SEM, XRD, FT-IR, and XPS analysis techniques. Besides diverse
morphology, both the polymeric materials were found to comprise similar chemical
natures bearing protonic acid–SO3H and Lewis base–C=N functionalities.
Subsequently, both the COPs were evaluated for the synthesis of
hydroxymethylfurfural (HMF) by the dehydration of fructose to investigate their
morphology-dependent catalytic activity.
60. Ratiometric pH-responsive 19F MRI contrast agents based on hydrazone switches
Dawid Janasik, Krzysztof Jasiński, Władysław Węglarz, Ivan Nemec, Paweł Jewuła, Tomasz Krawczyk, Anal. Chem., 2022 94, 8, 3427–3431, Citations
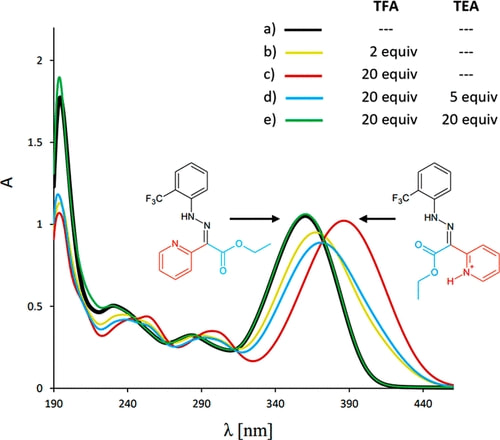
Hydrazone-based molecular switches serve as efficient ratiometric pH-sensitive agents that can be tracked with 19F NMR/MRI and 1H NMR. Structural changes induced between pH 3 and 4 lead to signal appearance and disappearance at 1H and 19F NMR spectra allowing ratiometric pH measurements. The most pronounced are resonances of the CF3 group shifted by 1.8 ppm with 19F NMR and a hydrazone proton shifted by 2 ppm with 1H NMR.
59. Trigonally distorted hexacoordinate Co(II) single-ion magnets
Ivan Nemec, Ondrej F. Fellner, Berenika Indruchova, Radovan Herchel, Materials, 2022, 15(3), 1064, Citations
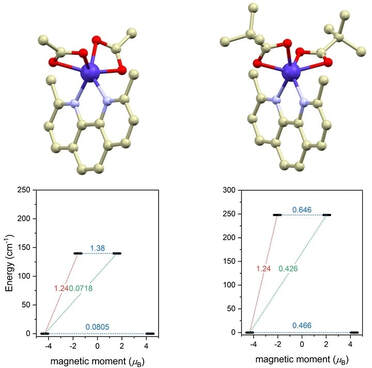
By simple reactions involving various cobalt(II) carboxylates (acetate and in situ prepared pivalate and 4-hydroxybenzoate salts) and neocuproine (neo), we were able to prepare three different carboxylate complexes with the general formula [Co(neo)(RCOO)2], (R = CH3 for 1, (CH3)3C for 2 and 4OH-C4H6 for 3). The [Co(neo)(RCOO)2] molecules in the crystal structures of 1-3 adopt rather distorted coordination environment with the largest trigonal distortion observed for 1, whereas 2 and 3 are distorted from ideal octahedral geometry similarly. The combined theoretical and experimental investigations of magnetic properties revealed that the spin Hamiltonian formalism was not a valid approach and the L-S Hamiltonian had to be used to reveal very large magnetic anisotropies for 1-3. The measurements of AC susceptibility showed that all three compounds exhibited slow-relaxation of magnetization in a weak external static magnetic field and thus can be classified as field induced single-ion magnets. It is noteworthy that 1 also exhibits a weak AC signal in a zero-external magnetic field.
2021
58. 3d-4f magnetic exchange interactions and anisotropy in a series of heterobimetallic vanadium(IV)-lanthanide(III) Schiff base complexes
Kamil Kotrle, Ivan Nemec, Jan Moncol, Erik Cizmar and Radovan Herchel , Dalton Trans. 2021, 50, 13883-13893, Citations
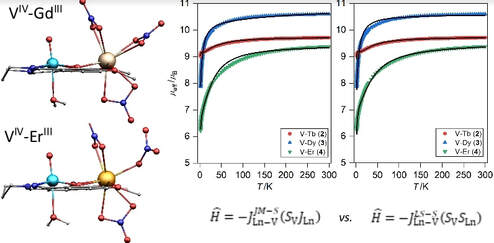
A series of heterobimetallic LnIII–VIV compounds [Ln(VO)L(NO3)3(H2O)] (Ln = Gd(1), Tb(2), Dy(3), Er(4)) assembled by Schiff base ligand (H2L = N,N′-bis(1-hydroxy-2-benzylidene-6-methoxy)-1,7-diamino-4-azaheptane) were prepared and studied with experimental and theoretical methods. The single-crystal X-ray analysis revealed the change of the coordination number from 10 found in 1-3 to 9 confirmed in 4. The DC magnetic data were fitted with several Hamiltonians to extract exchange and anisotropy parameters for all complexes 1-4. This investigation of magnetic properties was confronted with both DFT and CASSCF theoretical calculations. It was found out, that exchange interactions in 1, 3 and 4 are antiferromagnetic, while 2 has ferromagnetic exchange interaction. Moreover, AC susceptibility measurements revealed field-induced slow relaxation of magnetization in complexes 2 and 3 complicated by the presence of three relaxation channels. Nevertheless, these compounds belong to the first TbIII–VIV and DyIII–VIV single-molecule magnets in this class of compounds.
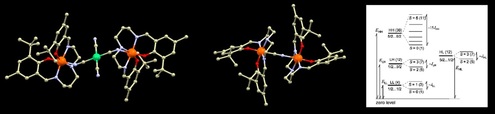
57.From Tetranuclear to Pentanuclear [Co‒Ln] (Ln = Gd, Tb, Dy, Ho) Complexes Across the Lanthanide Series: Effect of Varying Sequence of Ligand Addition
Dipmalya Basak, Lucy Smythe, Radovan Herchel, Mark Murrie, Ivan Nemec, Debashis Ray, Dalton Trans., 2021, 50, 11861-11877, Citations
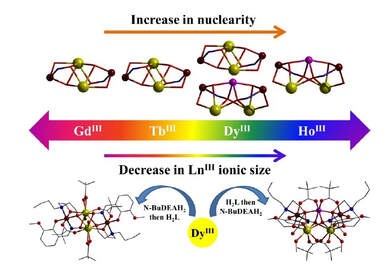
Two new families of cobalt(II/III)‒lanthanide(III) coordination aggregates have been reported:
Tetranuclear [LnIII
2CoIII
2L2(N‒BuDEA)2(O2CCMe3)4(H2O)2]·(MeOH)n·(H2O)m (Ln = Gd, 1; Tb,
2; Dy, 3; n=2, m=10 for 1 and 2; n=6, m=2 for 3) and Pentanuclear
LnIII
2CoIICoIII
2L2(N‒BuDEA)2(O2CCMe3)6(MeOH)2 (Ln = Dy, 4; Ho, 5) formed from the
reaction of two aggregation assisting ligands H2L (o‒vanillin oxime) and N‒BuDEAH2
(N‒butyldiethanolamine). A change in preference from lower to higher nuclearity structure was
observed on going across the lanthanide series brought about by the variation in size of the LnIII
ions. An interesting observation was made for the varying sequence of addition of the ligands in
the reaction medium paving the way to access both structural types for Ln = Dy. HRMS (+ve) of
solutions gave further insight into the formation of the aggregates via different pathways. The
tetranuclear complexes adopt a modified butterfly structure with a more complex bridging
network while trapping of an extra CoII ion in the pentanuclear complexes destroy this
arrangement putting the Co‒Co‒Co axis above the Ln‒Ln axis. Direct current (dc) magnetic
susceptibility measurements reveal weak antiferromagnetic coupling in 1. Complexes 2 and 5
display no slow magnetic relaxation, whereas complexes 3 and 4 display out‒of‒phase signals at low temperature. All compounds were analyzed with DFT and CASSCF calculations and
information about the single-ion anisotropies and mutual 4f‒4f / 4f‒3d magnetic interactions
were derived.
56. Solvent-induced structural transformation from heptanuclear to decanuclear [Co–Ln] coordination clusters: trapping of unique counteranion and understanding of aggregation pathways
Dipmalya Basak, Emma Regincós Martí, Mark Murrie, Ivan Nemec and Debashis Ray, Dalton Trans., 2021,50, 9574-9588. Citations
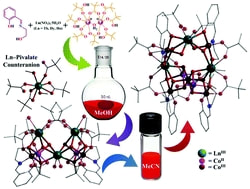
Five new cobalt(II/III)–lanthanide(III)-based coordination aggregates, [LnIII3CoII2CoIII2(L1)2(O2CCMe3)8(OH)4(OMe)2(H2O)4]·Ln(η1-O2CCMe3)2(η2-O2CCMe3)2(MeOH)2·2MeOH·2H2O (where Ln = Tb (1), Ho (3), and H2L1 = N-(2-hydroxyethyl)-salicylaldimine), TbIII3CoII3CoIII4(L1)4(O2CCMe3)9(OH)10(H2O) (4) and LnIII3CoII2CoIII5(L1)4(O2CCMe3)10(OH)10 (Ln = Dy (5), Ho (6)) have been synthesized and characterized, including structural analysis via single-crystal X-ray diffraction. The dysprosium analogue (2) of 1 and 3 was previously reported by us. The heptanuclear monocationic clusters in 1 and 3 were formed by placement of seven metal ions (4 Co and 3 Ln) in a vertex shared dicubane structure from the control of two Schiff base anions and crystallized in the presence of in situ generated and literature unknown counter anions Tb(η1-O2CCMe3)2(η2-O2CCMe3)2(MeOH)2− and Ho(η1-O2CCMe3)2(η2-O2CCMe3)2(MeOH)2−. Interesting solvent-induced cluster structure transformation was observed on dissolving the heptanuclear aggregates in MeCN for the formation of decanuclear clusters 4–6. These high nuclearity clusters consist of a vertex shared heptanuclear dicubane part and a curved trinuclear chain linking the two cubic halves. The dicubane unit differs from that of the heptanuclear precursors in the presence of CoII/III at the shared vertex as opposed to LnIII and the absence of OMe− bridges. HRMS (+ve) analysis shed light on the pathway of formation of these heptanuclear molecules, while at the same time revealing a different aggregation process for the decanuclear clusters.
55. Unexpected solution behaviour of ester-functionalized half-sandwich Ru(II) and Ir(III) complexes.
Lukáš Masaryk, Ivan Nemec, Jana Kasparkova, Viktor Brabec and Pavel Starha, Dalton Trans. 2021, 50, 8017-8028, Citations
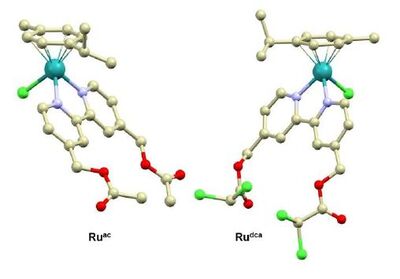
Complexes [Ru(η6-pcym)(bpydca)Cl]PF6 (Rudca) and [Ir(η5-Cp*)(bpydca)Cl]PF6 (Irdca) were developed as model compounds for the investigation of multi-targeted ester-functionalized half-sandwich ruthenium(II) and iridium(III) complexes; pcym = 1-methyl-4-(propan-2-yl)benzene (p-cymene), bpydca = 2,2'-bipyridine-4,4'-diyldimethanediyl bis(dichloroacetate), Cp* = pentamethylcyclopentadienyl. Aiming to understand the in-solution behaviour of these first-in-class complexes containing the pyruvate dehydrogenase kinase inhibitor dichloroacetate (dca) as the terminal bioactive substituent, several experiments were performed under aqueous conditions for Rudca and Irdca, as well as for compounds [Ru(η6-pcym)(bpyOH)Cl]PF6 (RuOH) and [Ir(η5-Cp*)(bpyOH)Cl]PF6 (IrOH), and acetyl analogues [Ru(η6-pcym)(bpyac)Cl]PF6 (Ruac) and [Ir(η5-Cp*)(bpyac)Cl]PF6 (Irac) bearing a different (biologically inactive) terminal substituent; bpyOH = 2,2'-bipyridine-4,4'-diyldimethanol, bpyac = 2,2'-bipyridine-4,4'-diyldimethanediyl diacetate. The experiments were also conducted in the presence of porcine liver esterase (PLE). All the six complexes were characterized by relevant techniques (e.g., NMR and mass spectrometry), including a single-crystal X-ray analysis of complexes Rudca, RuOH, IrOH and Ruac. Although designed as model compounds, complexes Rudca, Irdca, RuOH and IrOH were also screened for their antiproliferative activity in four human cancer cell lines (HCT116 colon carcinoma, MDA-MB-231 and MCF-7 breast adenocarcinomas, DU145 prostate carcinoma), where the tested complexes did not show any effect (IC50 > 100 μM).
54. Hydroxido Supported and Differently Networked Octanuclear Ni6Ln2 [Ln = GdIII and DyIII] Complexes: Structural Variation, Magnetic Properties and Theoretical Insights
Avik Bhanja, Lucy Smythe, Radovan Herchel, Ivan Nemec, Mark Murrie and Debashis Ray, Dalton Trans., 2021, 50, 5023. Citations
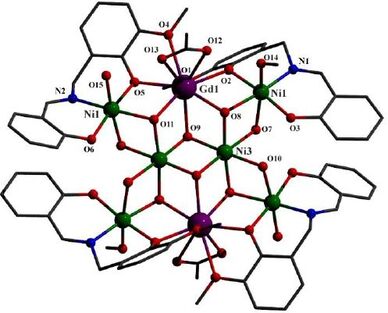
The design and synthesis of Schiff base H2L, (2-{[(2-hydroxy-3-methoxybenzyl)imino]methyl}phenol) bearing ONOO donor set has been explored for its reactivity pattern with LnCl3∙6H2O (Ln = GdIII and DyIII) and Ni(CH3CO2)2∙4H2O in the presence of NEt3 for molecular aggregation. Coordination driven spontaneous self-assembly reactions provides [Gd2Ni6L4(μ3-OH)6(μ-OH)2(μ-H2O)2(CH3CO2)2(H2O)3(MeOH)]∙8H2O (1), having ‘butterfly−shaped’ core and [Dy2Ni6L4(μ3-OH)4(μ-Cl)2Cl4(H2O)2(MeOH)2]∙2MeOH∙4H2O (2) with ‘candy–shaped’ core. Fusion of six partial cubane units led to mineral-like core in 1, developed around central Ni2(OH)2 moiety; whereas in case of 2, four such partial cubanes are collapsed on Dy2(OH)2. Direct-current (dc) magnetic susceptibility measurements reveal predominantly ferromagnetic interactions leading to a high‒spin S = 13 ground state for 1. Complex 2 exhibits slow relaxation of magnetization in a small applied dc field with an energy barrier to reorientation of the magnetization, Ueff = 19.3 (0.1) K. The static and dynamic magnetic data are analysed and corroborated by density functional theory (DFT) and detailed CASSCF based calculations.
53. Dinuclear half-sandwich Ir(III) complexes containing 4,4'-methylenedianiline-based ligands: synthesis, characterization, cytotoxicity
Lukáš Masaryk, Karolina Słoczyńska, David Milde, Ivan Nemec, Paulina Koczurkiewicz-Adamczyk, Elżbieta Pękala, Pavel Štarha, J. Organometallic Chemistry, 2021, Volume 938, 121748, Citations
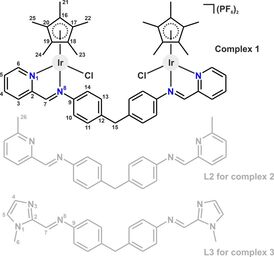
Three dinuclear half-sandwich iridium(III) complexes [Ir2(μ-L1)(η5-Cp*)2Cl2](PF6)2 (1), [Ir2(μ-L2)(η5-Cp*)2Cl2](PF6)2 (2) and [Ir2(μ-L3)(η5-Cp*)2Cl2](PF6)2 (3) with tetradentate 4,4'-methylenedianiline-based N-donor ligands (L1–L3) were prepared and characterized by NMR spectroscopy, elemental analysis, mass spectrometry and FTIR spectroscopy. Complex 3 was hydrolytically less stable in 20% DMSO-d6/80% D2O than 1 and 2, while all the studied complexes were stable in the same mixture of solvents in the presence of PBS (pH 7.4). In addition, complexes 1–3 converted coenzyme NADH to its oxidized form (NAD+). Cytotoxicity of 1–3 was evaluated on five human cancer cell lines, in particular lung carcinoma (A549), hepatocellular carcinoma (HepG2), melanoma (A375), prostate carcinoma (DU-145) and breast carcinoma (MCF-7). The best-performing complex 2 showed comparable cytotoxicity to conventional cisplatin against the A549 and HepG2 cell lines.
52. А new series of Schiff base Ni(II)4 cubanes: Evaluation of magnetic coupling via carboxylate bridges
Alexey N Gusev, Ivan Nemec, Radovan Herchel, Yuriy I Baluda, Maria A Kryukova, Nikolay N Yefimov, Mikhail A. Kiskin, Wolfgang Linert, Polyhedron, 2021, Volume 196, 115017
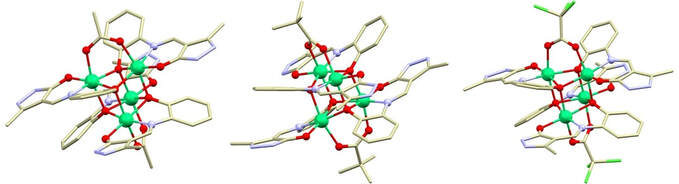
The Schiff base ligand H2L has been synthesized by condensation of 2-aminophenol with 3-methyl-1-phenyl-4-formylpyrazol-5-one and reaction between H2L and Ni(OOCR3)2, R = H in 1, -CH3 in 2, Cl in 3, yielded three new tetranuclear Ni(II) complexes. The complexes have been characterized by elemental analysis, IR- and ES-MS spectroscopy. Their structures as well as structure of ligand were determined by single-crystal X-ray diffraction. Compounds 1-3 possesses tetranuclear cubane-like structures containing [Ni4L4(R3COO)2]2- complex anions, which are charge balanced by two triethyl ammonium cations. Furthermore, the crystal structure of the Ni(II) cubane compound containing tri-chloroacetate bridging ligands, is reported for the first time. Variable temperature magnetic suscep-tibility measurements revealed interplay between ferromagnetic and antiferromagnetic exchange in the tetranuclear cubane-like compounds 1-3, in which ferromagnetic interactions were enhanced by introducing carboxylate bridging groups. DFT calculations supported the analysis of magnetic data
2020
51. Deposition of Tetracoordinate Co(II) Complex with Chalcone Ligands on Graphene
Jakub Hrubý, Šárka Vavrečková, Lukáš Masaryk, Antonín Sojka, Jorge Navarro-Giraldo, Miroslav Bartoš, Radovan Herchel, Ján Moncol, Ivan Nemec and Petr Neugebauer, Molecules. Spec. issue: "Hybrid Materials Advanced Applications", 2020, 25(21), 5021. Citations
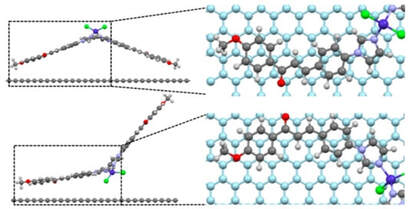
Studying the properties of complex molecules on surfaces is still mostly an unexplored research area because the deposition of the metal complexes has many pitfalls. Herein, we probed the possibility to produce surface hybrids by depositing a Co(II)-based complex with chalcone ligands on chemical vapor deposition (CVD)-grown graphene by a wet-chemistry approach and by thermal sublimation under high vacuum. Samples were characterized by high-frequency electron spin resonance (HF-ESR), XPS, Raman spectroscopy, atomic force microscopy (AFM), and optical microscopy, supported with density functional theory (DFT) and complete active space self-consistent field (CASSCF)/N-electron valence second-order perturbation theory (NEVPT2) calculations. This compound’s rationale is its structure, with several aromatic rings for weak binding and possible favorable π–π stacking onto graphene. In contrast to expectations, we observed the formation of nanodroplets on graphene for a drop-cast sample and microcrystallites localized at grain boundaries and defects after thermal sublimation.
50. Co(II)-based Single-Ion Magnets with 1,1′-ferrocenediyl-bis(diphenylphosphine) metalloligands
J. Hrubý, D. Dvořák, L. Squillantini, M. Mannini, J. van Slageren, R. Herchel, I. Nemec, P. Neugebauer, Dalton Trans. 2020, 49, 11697-11707. Citations
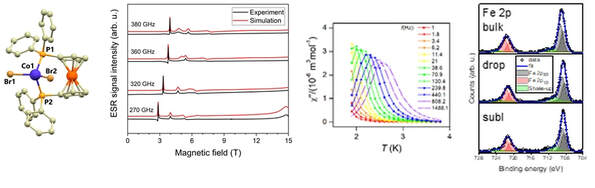
we report on investigations of magnetic and spectroscopic properties of three heterobimetallic Fe(II)-Co(II) coordination compounds based on the tetracoordinate {CoP2X2} core encapsulated by dppf metalloligand, where X = Cl (1), Br (2), I (3), dppf = 1,1′-ferrocenediyl -bis(diphenylphosphine). The analysis of static magnetic data has revealed the presence of axial magnetic anisotropy in compounds (1) and (2) and this was further confirmed by high-frequency electron spin resonance (HF-ESR) spectroscopy. Dynamic magnetic data confirmed that (1) and (2) behave as field-induced Single-Ion Magnets (SIMs). Together with bulk studies, we have also tested the possibility of depositing (2) as thick films on Au(111), glass, and polymeric acetate by drop-casting as well as thermal sublimation, a key aspect for the development of future devices embedding these magnetic objects.
49. Halogen bonding in new dichloride-cobalt(II) complex with iodo-substituted chalcone ligands
L. Masaryk, J. Moncol, R. Herchel and I. Nemec, Crystals 2020, 10(5), 354. Citations
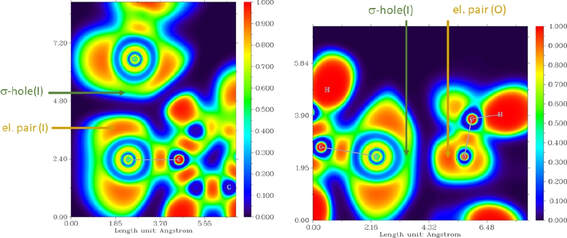
The synthesis and properties of new chalcone ligand 4I-L ((2E)-1-[4-(1H-imidazol-1-
yl)phenyl]-3-(4-iodophenyl)prop-2-en-1-one) and tetracoordinate Co(II) complex [Co(4I-L)2Cl2],
(1a), are reported in this article. Upon recrystallization of 1a the single crystals of [Co(4I16
L)4Cl2]·2DMF·3Et2O, (1b) were obtained and crystal structure was determined using X-ray
diffraction. The non-covalent interactions in 1b were thoroughly analyzed and special attention was
dedicated to interactions formed by the peripheral iodine substituents. The density functional
theory (DFT), atoms in molecule (AIM) and noncovalent interaction (NCI) methods and electronic
localization function (ELF) calculations were used to investigate halogen bond formed between the
iodine functional groups and co-crystallized molecules of diethyl ether.
48. The effect of coordination geometry on magnetic properties in a series of cobalt(II) complexes and structural transformation in mother liquor
S. Ghosh, S. Kamilya, M. Das, M. Sakshi; E.M. Boulon, I. Nemec, M. Rouzieres, R. Herchel, A. Mondal, Inorg. Chem., 2020, 59, 10, 7067–7081. Citations
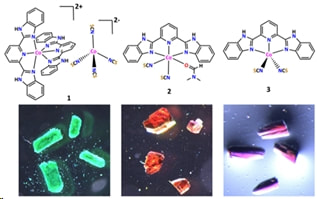
Three Co(II) complexes [Co(bbp)2][Co(NCS)4]•4DMF (1), [Co(bbp)(NCS)2(DMF)]•2DMF (2) and [Co(bbp)(NCS)2] (3) have been synthesized and characterized by single-crystal X-ray diffraction, magnetic and various spectroscopic techniques. Complexes 1 and 3 are obtained by the reaction of Co(NCS)2 with 2,6-bis(1H-benzo[d]imidazol-2-yl)pyridine (bbp) whereas complex 1 undergo a crystal to crystal transformation to form complex 2. Single-crystal X-ray revealed that complex 1 comprises of two Co(II) centers: a cationic octahedral Co(II) unit and an anionic tetrahedral Co(II) unit, while Co(II) ion is in distorted octahedral environment in 2. Moreover, in complex 3, Co(II) ion is in distorted square pyramidal geometry. The impact of coordination geometry on the magnetic property was studied both by static and dynamic magnetic measurements. Direct current (dc) magnetic susceptibility measurements showed that all the Co(II) ions are in high-spin state in these complexes. Alternating current (ac) magnetic susceptibility measurements indicated that the complexes 2 and 3 display slow relaxation of magnetization in an external dc magnetic field, while complex 1 displayed no such property. EPR experiments and theoretical calculations were consistent with the above findings.
47. Heteronuclear Iron(III)–Schiff Base Complexes with the Hexacyanidocobaltate(III) Anion: On the Quest To Understand the Governing Factors of Spin Crossover
Ján Pavlik, Petra Masárová, Ivan Nemec, Olaf Fuhr, Mario Ruben, Ivan Šalitroš, Inorg. Chem., 2020, 59, 5, 2747-2757. Citations
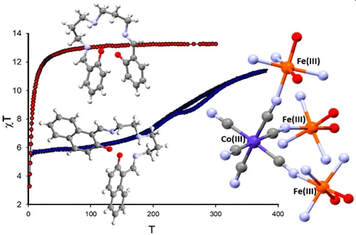
Two heteronuclear compounds (1 and 2) containing three ferric centers linked in facial-like mode with the magnetically silent hexacyanidocobaltate(III) anion were prepared and studied. The structural investigation revealed that both compounds are tetranuclear complexes with molecular formulas of [{Fe(L1)NC}3Co(CN)3]·2CH3OH·2.5CH3CN (1) and [{Fe(L2)NC}3Co(CN)3]·2H2O·1CH3OH (2). The magnetic properties of both complexes are controlled by the molecular design of the corresponding pentadentate Schiff base anions L12– and L22–. While compound 2 with a symmetric ligand prepared from salicylaldehyde shows high-spin state properties, compound 1 containing the asymmetric ligand with naphthalene units either is low-spin in its solvated form or shows a gradual but hysteretic spin crossover event when desolvated. The magnetic behavior was analyzed with respect to the Ising-like model and spin Hamiltonian, respectively, and the results were confronted with ab initio calculations. Additionally, the influence of structural features, lattice solvent molecules, the distribution of electronic terms, and active orbitals on the spin state properties of reported complexes is discussed.
46. Nanostructured graphene for nanoscale electron paramagnetic resonance spectroscopy.
Luke St. Marie, Abdel El Fatimy, Jakub Hrubý, Ivan Nemec, James Hunt, Rachael L. Myers-Ward, Kurt Gaskill, Mattias Kruskopf, Yanfei Yang, R E Elmquist, J.Phys. Materials, 2020, Vol. 3, No. 1, 014013
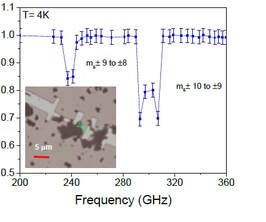
The opening of a quantum confinement gap in nanostructured graphene yields extremely sensitive photodetectors, with electrical noise equivalent power lower than 10-15 W Hz-0.5 at temperatures below 3 K, for detection of radiation in a very broad frequency range, including ultraviolet, visible and terahertz. Here we demonstrate the operation of these detectors in the presence of magnetic field as high as 7 T, paving the way to in situ spectroscopy of molecular nanomagnets.
2019
45. Spin crossover in three mononuclear iron (III) Schiff-base complexes
Ivan Nemec, Ingrid Svoboda and Radovan Herchel, Metals, Special issue: "Molecular Magnetism of Transition Metal Complexes", 2019, 9(8), 849. Citations.
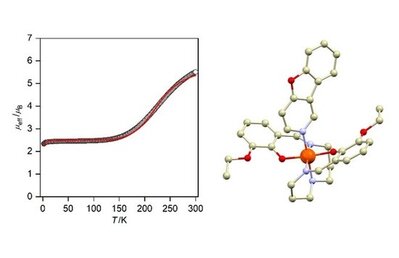
The synthesis, crystal structure, and magnetic properties of three new mononuclear complexes [Fe(R-LA)(L1)](BPh4), where R-LA2− is a doubly deprotonated pentadentate Schiff base ligand and L1 is a monodentate benzimidazole or furopyridine ligand, are reported. Ligand- and anion-driven changes in crystal structures and magnetic behavior were investigated in terms of the magnetic susceptibility measurements and theoretical calculations.
44. Impact of the Schiff Base Ligand Substituents on the Solid State and Solution Properties in Eleven Iron(III) Complexes
Lukáš Pogány, Barbora Brachnakova, Petra Masarova, Jan Moncol, Jan Pavlik, Miroslav Gal, Milan Mazur, Radovan Herchel, Ivan Nemec and Ivan Salitros , New J. Chem., 2019, 43 (35), 13916-13928. CItations
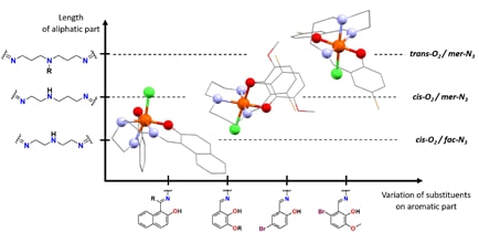
Investigations on a series of mononuclear iron(III) Schiff base complexes of general formula [Fe(L)Cl]·S (where L2- is Schiff base ligand anion, S is a solvent molecule) are reported. Derivatives of salicylaldehyde or 2-hydroxy-1-naphtaldehyde were used in combination with a linear either symmetrical or non-symmetrical aliphatic triamines to synthesize the Schiff base ligands and the consecutive reactions with iron(III) chloride afforded various [Fe(L)Cl] complexes. Compounds were characterized by conventional techniques and the crystal structures of all complexes have been determined as well. Structural study has revealed relationship between the spatial arrangement of N3O2-donor atom set and length of the aliphatic part of Schiff base ligand. Magnetic and EPR investigation confirmed the high spin state behaviour in all reported compounds and their analysis allowed to quantify the parameters of spin Hamiltonian. Formation of hydrogen-bonded pseudodimers with antiferromagnetic exchange coupling was found in the complexes with the shortest aliphatic part of Schiff base ligands. Experimental exchange coupling constants are in good agreement with those obtained from DFT calculation and correlate with the strength of the hydrogen bonds. Cyclic and square wave voltammetry were employed to investigate the redox properties of reported compounds and the experimental redox potentials were compared with those obtained from the DFT calculations.
43. Structural and magnetic characterization of Ni(II), Co(II), Fe(II) binuclear complexes on bis(pyridyl-triazolyl)alkane basis.
Alexey Gusev, Ivan Nemec, Radovan Herchel, Irina Riush, Ján Titiš, Roman Boča, Konstantin Lyssenko, Mikhail Kiskin, Igor Eremenko, Wolfgang Linert, Dalton Trans., 2019, 48, 10526-10536.
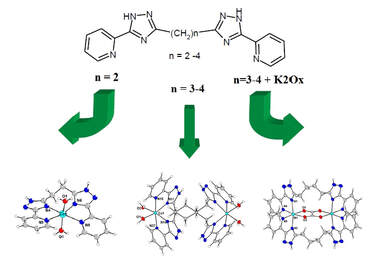
Reaction of bis[5-(2-pyridyl)-1,2,4-triazol-3-yl]alkanes (alkane spacers = -CH2- in L2, -C3H6- in L3, -C4H8- in L4) with M(II)A2
salts (M = Ni, Co, Fe) resulted in preparation of five series of mononuclear ([M(L2)(H2O)2]2+, 1a-c) or binuclear
([M2(L3)2(H2O)4]4+, 2a-c; ([M2(L4)2(H2O)4]4+, 3a-c, [M2(L3)2(-ox)]2+, 4a-c; [M2(L4)2(-ox)]2+, 5a-c) complexes. The crystal
structures of ten complexes were determined by single-crystal X-ray crystallography. Magnetic properties of the
compounds were characterized by SQUID magnetometry and were analyzed by fitting on spin Hamiltonian model . It was
revealed that Fe(II) and Co(II) compounds exhibit non-negligible anisotropy and in the case of 2a-c and 3a-c complexes
weak ferromagnetic interactions between the metal centers were observed. In the case of complexes containing {M2(-
ox)}2+ core strong antiferromagnetic interactions were observed within the dimer. Remarakably, solid state luminiscence
of Co(II) and Fe(II) complexes (1b, 2b, 3b and 1c, 2c, 3c) was observed.
42. Ion-pair complexes of Schiff base Fe(III) cations and complex anions.
Ivan Nemec, Pavel Zoufalý, Pawel Jewula, Peter Antal, Wolfgang Linert, and Radovan Herchel, New J. Chem., 2019, 43, 4937 - 4946. Citations.
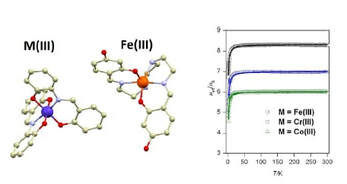
Compound [Fe(H2-4OH-L6)]Cl was used for preparation of four new ion-pair complexes with general formula: [Fe(H2-4OHL6)][
M(L3)2]·H2O (M = CoIII, (3a), CrIII
, (3b) and FeIII, (3c)) and [Fe(H2-4OH-L6)][Ag(CN)2], (3d), where H4-4OH-L6 = N,N'-Bis[2,4-
dihydroxy-(benzylideneamino)ethyl]ethane-1,2-diamine and H2L3 = 2-{(E)-[(2-hydroxyphenyl)imino]methyl}phenol.
Furthermore, two [Fe(H-4OH-L6)] complexes (1a and 1b) with monodeprotonated hexadentate ligand were prepared. The
crystal structures were determined by single-crystal X-ray measurements for all the above-mentioned complexes excluding
3c. Its isostructurality with 3a-b was confirmed by powder X-ray diffraction. The magnetic properties were investigated by
static magnetic measurements and they are dominated by the high spin ground state of the complex cations and anions
and at smaller scale by the intermolecular interactions presented among the molecules.
41. Copper(II)) self-assembled clusters of bis((pyridin-2-yl)-1,2,4-triazol-3-yl)alkanes. Unusual rearrangement of ligand upon reaction condition.
Alexey Gusev, Ivan Nemec, Radovan Herchel, Victor Shul’gin, Irina Ryush, Michail Kiskin, Nickolay Efimov, Elena Ugolkova, Vadim Minin, Konstantin Lyssenko, Igor Eremenko, Wolfgang Linert, Dalton Trans., 2019, 48, 3052-3060. Citations.
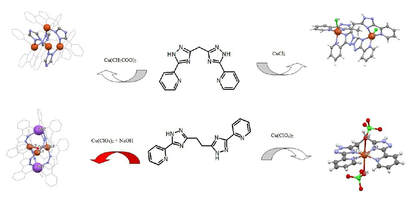
Reaction of two structurally related bridging ligands bis[5-(2-pyridyl)-1,2,4-triazole-3-yl]methane (H2L1) and bis[5-(2-
pyridyl)-1,2,4-triazole-3-yl]ethane (H2L2) with copper(II) salts resulted in a surprising wide variety of the complex
structures [Cu2(H2L1)Cl2]Cl2·4CH3OH (1), [Cu4L14]∙4H2O (2), [Cu(H2L2)(ClO4)2] (3) and [Cu3(OH)Na2(L’)6](ClO4)·5H2O·C3H6O (4),
where HL' is 3,5-bis-(pyridin-2-yl)-1,2,4-triazole, which were structurally characterized by the X-ray diffraction method.
Complexes 1 and 2 were prepared on H2L1 basis and have binuclear and tetranuclear structure respectively,
demonstrating strong impact of type of counter anion on coordination mode of the ligand. In contrast, the reaction
between Cu(ClO4)2 6H2O and H2L2 led to preparation of mononuclear complex 3. Reaction of H2L2 with Cu(ClO4)2 in alkaline
conditions led to oxidative rearrangement of the ligand and the homoleptic pentanuclear complex 4 with anionic ligand L’
was prepared. Magnetic properties were studied for compounds 1, 2 and 4 and for all of them the antiferromagnetic
interactions between the Cu atoms were confirmed and analyzed by spin Hamiltonian formalism. Furthermore, the
occurrence of the antisymmetric exchange was confirmed in 4. The magnetic data analysis was supported by the X-band
EPR measurements performed for complexes 1, 2 and 4.
40. The effect of the second coordination sphere on the magnetism of [Ln(NO3)3(H2O)3]·(18-crown-6) (Ln = Dy and Er)
Radovan Herchel, Pavel Zoufalý and Ivan Nemec, RSC Adv. 2019, 9, 569-575. Citations.
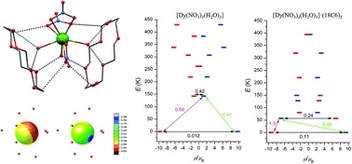
The objective of this work was the exploration of the effect of the second coordination sphere on the magnetic properties of [Ln(NO3)3(H2O)3]·(18C6) (Ln = Dy (1) and Er (2)) compounds comprising co-crystallized 18-crown-6 ethers. Both compounds were identified as field-induced single molecule magnets (SMMs) with estimated magnetization reversal barriers Ueff = 66–71 K for 1 and Ueff = 21–24 K for 2. Theoretical calculations with the B3LYP functional revealed substantial change and redistribution of the electrostatic potential upon accounting for the second coordination sphere represented by two 18C6 molecules, which resulted in the change of the crystal-field around metal atoms. As a result, the multireference CASSCF calculations exposed significant impact of the second coordination sphere on the energy splitting of the respective 6H15/2 (DyIII) and 4I15/2 (ErIII) ground states, the magnetization reversal barrier and the magnetic anisotropy parameters.
2018
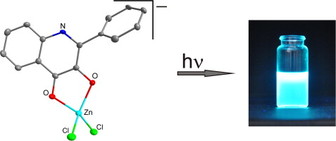
39. Synthesis and photophysical properties of Zn(II) Schiff base complexes possessing strong solvent-dependent solid-state fluorescence
A.N.Gusev, F.Shulgin, E.V.Braga, I.Nemec, F.Minaev, G.V.Baryshnikov, Z.Trávníček, H.Ågren, I.L.Eremenko, K.A.Lyssenko, W.Linert, Polyhedron, 2018, 155, 202-208. Citations.
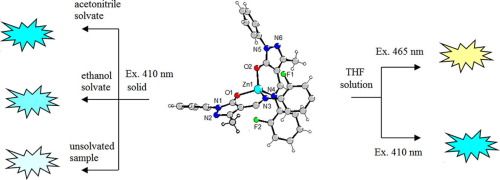
The present article reports on the syntheses, crystal structures and luminescence properties of three solvate forms of a zinc(II) complex containing 4-{(E)-[(2-fluorophenyl)imino]methyl}-5-methyl-2-phenyl-2,4-dihydro-3H-pyrazol-3-one (HL). The reaction of zinc(II) acetate with the HL ligand in ethanol and acetonitrile led to the formation of two solvate analogues [Zn(L)2]·Solv (Solv – ethanol (1) and acetonitrile (2)). The properties of the [Zn(L)2]·Solv complexes were investigated by UV-Vis absorption and fluorescence emission spectroscopy, and density functional theory calculations. Bader’s topological analysis was performed to investigate the electronic peculiarities of Zn(II) polyhedra and non-covalent interactions within crystal packing of studied solvates.
38. Impact of the Substituent Variation on the Presence of Thermal Spin Crossover in the Series of Mononuclear Iron(III) Schiff Base Complexes with Terminal Pseudohalido Coligands
Ivan Salitros, Lukáš Pogány, Barbora Brachňaková, Jan Moncol, Jan Pavlik, Ivan Nemec, Zdeněk Trávniček, Milan Mazúr, Lukáš Bučinský, Ľubomír Suchánek, Chemistry-A European Journal, 2018, Vol. 24, Iss.20, 5191-5203. Citations.
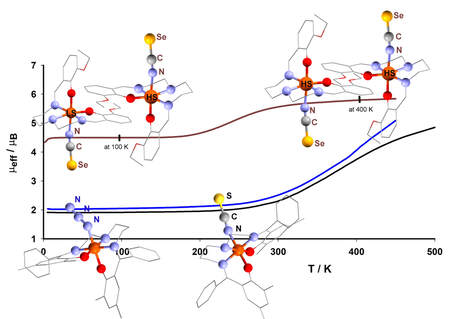
A series of novel iron(III) complexes of the general formula [Fe(L)X] (where L stands for a dianion of
pentadentate Schiff base ligand N,N’-bis((2-hydroxy-3,5-dimethylphenyl)phenyl)methylidene-1,6-
diamino-3-azapentane=H2L1 for 1 and 2; N,N’-bis((2-hydroxy-3-ethoxyphenyl)methylidene)-1,6-
diamino-3-azapentane=H2L2 for 3 and 3·C3H6O) and X is terminal pseudohalido ligand (X=N3 for 1,
X=NCS for 2, and X=NCSe for 3 and 3·C3H6O) were synthesized and thoroughly characterized.
Magnetic measurements revealed the above room temperature spin crossover for isomorphic
complexes 1 and 2 (T1/2 = 441 K and T1/2 = 435 K, respectively), while the solvent-free complex 3
showed a half complete spin crossover (T1/2 = 250 K), which was detected by a variable temperature
crystallography as well. On the other hand, solvated complex 3·C3H6O exhibited permanent high spin
state behaviour and either recrystallization or in situ thermal desolvation converts 3·C3H6O to
solvent-free and spin crossover active form 3. Magnetic properties of all the reported complexes
were also supported by EPR spectroscopy experiments and in addition, DFT and ab initio calculations
were employed for the evaluation of the g-factor and ZFS parameters.
37. Two polymorphic Co(II) field-induced single-ion magnets with enormous angular distortion from the ideal octahedron
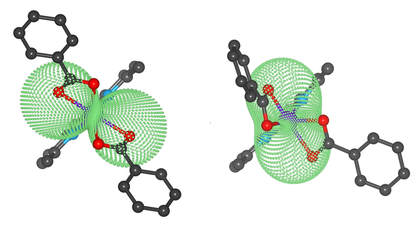
A mononuclear complex [Co(neo)(PhCOO)2,], neo = neocuproine, PhCOO- = the benzoate anion, was prepared in two polymorph forms crystallizing in the C2/c, (1) and P21/c, (2) space groups. The polymorphs differ in the Co-O bond lengths and the level of trigonal distortion of their coordination polyhedra. The static and dynamic magnetic properties of these compounds were thoroughly studied by experimental (magnetometry) and theoretical (ab initio calculations) methods. The analysis of magnetic data was performed using the spin Hamiltonian formalism or L-S model considering also the orbital angular momentum. It was revealed that both polymorphs possess a very large magnetic anisotropy with a pronounced rhombic character leading to the separation of the Kramers doublets larger than 120 cm-1. The measurements of alternating current susceptibility revealed that both polymorphs behave as field induced single molecule magnets with a small barrier of spin reversal (U = 22.1 K (for 1) and 17.1 K (for 2)) which indicates that other relaxation processes than the thermally activated Orbach process take place.
2017
36. Crystal structures and magnetic properties of two series of phenoxo-O bridged dinuclear Ln2 (Ln = Gd, Tb, Dy) complexes.
Marek Machata, Ivan Nemec, Radovan Herchel, Zdenek Travnicek, Dalton Trans., 2017, 46, 46, 16294-16305. Citations.
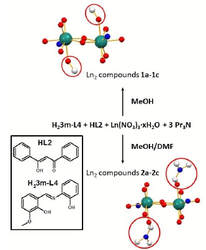
Six dinuclear lanthanide compounds of the formulas [Ln2(3m-L4)2(L2)2(MeOH)2]•6MeOH (Ln = Gd – 1a, Tb – 1b, Dy – 1c) and [Ln2(3m-L4)2(L2)2(DMF)2] (Ln = Gd – 2a, Tb – 2b, Dy – 2c; DMF = N,N-dimethylformamide, H23m-L4 = (2-[(E)-(3-metoxysalicylidene)amino]phenol), HL2 = 1,3-diphenyl-1,3-diketopropane) were prepared and characterized by elemental analysis, FTIR spectroscopy and thermogravimetric measurements, single-crystal X-ray structural analysis, magnetometry, and the Gd2 and Dy2 compounds by ab initio methods as well. The structural analysis revealed isostructurallity of the compounds within the series of 1a-c and 2a-c. Analysis of the variable temperature magnetic data showed the presence of a weak antiferromagnetic coupling in the Gd2 compounds (J/cm-1 = –0.13 for 1a and J/cm-1 = –0.17 for 2a). Magnetocaloric effect was studied on the compound 2a with the maximum value of –ΔSM = 22.9 J kg-1 K-1 at T = 2.0 K and B = 9 T, which is the highest value among the Gd2 double phenoxo-bridged compounds observed up to now. Both the Dy2 compounds (1c and 2c) exhibit slow-relaxation of magnetization in zero external static magnetic field. Magnetic anisotropy, intradimer magnetic coupling and magnetization blocking barrier were also studied by ab initio methods for 1c and 2c.
35. Tetranuclear Ni(II) and Co(II) Schiff-base complexes with an M4O6 defective dicubane-like core: zero-field SMM behavior in the cobalt analogue
Ivan Nemec, Radovan Herchel, Marek Machata, Zdenek Travnicek, New J., Chem., 2017, 41, 19, 11258-11267. Citations.
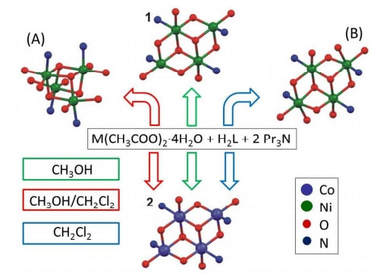
Two isostructural tetranuclear M(II)4 complexes with the general formula [M4(L)4(CH3OH)2] were prepared by the reaction
of M(CH3COO)2·4H2O and H2L in the presence of Pr3N (M = Ni in 1, Co in 2, H2L =2-{(E)-[(2-
hydroxyphenyl)imino]methyl}phenol, Pr3N = tripropylamine). The crystal structure of 1 was determined by single-crystal Xray
diffraction and it was revealed that it possesses the defective dicubane {Ni4O6}
2- core with two penta- and two
hexacoordinate Ni atoms. The isostructurality of both complexes was confirmed by powder X-ray diffraction. The analysis
of the static magnetic data revealed that the prevailing antiferromagnetic interaction leads to the diamagnetic ground
state in 1, whereas the ferromagnetic interactions dominate in 2. The analysis of magnetic data was supported by brokensymmetry
DFT and CASSCF/NEVPT2 calculations, where the latter disclosed large magnetic anisotropy in both compounds.
Moreover, the measurements of ac susceptibility in zero-applied magnetic field confirmed the presence of slow-relaxation
of magnetization in 2 and thus, this compound behaves as a single-molecule magnet.
34. An octanuclear Schiff-base complex with a Na2Ni6 core: structure, magnetism and DFT calculations
Marek Machata, Ivan Nemec, Radovan Herchel and Zdeněk Trávníček, RSC Adv., 2017,7, 25821-25827. Citations.
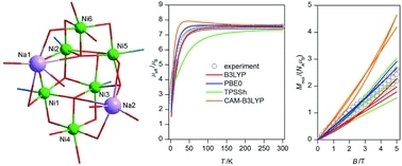
The octanuclear Na2Ni6 complex (Pr3NH)[Na2Ni6(L)4(Bza)5(HBza)(OH)2(ace)]·Et2O (1), where Pr3NH+ = the tripropylamonium cation, H2L = 2-[(E)-(2-hydroxybenzylidene)amino]phenol, HBza = benzoic acid and ace = acetone, was synthesized and characterized by the elemental analysis, FTIR spectroscopy, single crystal X-ray diffraction analysis, magnetic measurements and DFT calculations. All six NiII atoms are hexacoordinate with the {NiO6} or {NiNO5} chromophores forming two defective dicubane cores. The static magnetic data were fitted to the simplified spin Hamiltonian model which resulted in averaged ferromagnetic and antiferromagnetic exchange parameters J = +5.3 cm−1, and J = −9.2 cm−1, respectively, confirming the predominant role of the antiferromagnetic coupling in 1. The broken symmetry DFT method with various functionals (B3LYP, PBE0, TPPSh and CAM-B3LYP) was used to dissect information about magnetic coupling. As a result, the isotropic exchange parameters (Jab) derived by the PBE0 or B3LYP functionals seem to be the best to match the experimental magnetic data.
33. Magnetic Anisotropy and Field‐induced Slow Relaxation of Magnetization in Tetracoordinate Co(II) Compound [Co(CH3‐im)2Cl2]
Ivan Nemec, Radovan Herchel, Michal Kern, Petr Neugebauer, Joris van Slageren and Zdeněk Trávníček, Materials 2017, 10(3), 249. Citations.
Static and dynamic magnetic properties of the tetracoordinate CoII complex [Co(CH3‐im)2Cl2], (1, CH3‐im = N‐methyl‐imidazole), studied using thorough analyses of magnetometry, and High‐Frequency and ‐Field EPR (HFEPR) measurements, are reported. The study was supported by ab initio complete active space self‐consistent field (CASSCF) calculations. It has been revealed that 1 possesses a large magnetic anisotropy with a large rhombicity (magnetometry: D = −13.5 cm−1, E/D = 0.33; HFEPR: D = −14.5(1) cm−1, E/D = 0.16(1)). These experimental results agree well with the theoretical calculations (D = −11.2 cm−1, E/D = 0.18). Furthermore, it has been revealed that 1 behaves as a field‐induced single‐ion magnet with a relatively large spin‐reversal barrier (Ueff = 33.5 K). The influence of the Cl–Co–Cl angle on magnetic anisotropy parameters was evaluated using the CASSCF calculations.
2016
32. Pentacoordinate and Hexacoordinate Mn(III) Complexes of Tetradentate Schiff-Base Ligands Containing Tetracyanidoplatinate(II) Bridges and Revealing Uniaxial Magnetic Anisotropy
Ivan Nemec, Radovan Herchel and Zdeněk Trávníček, Molecules 2016, 21(12), 1681. Citations.
Crystal structures and magnetic properties of polymeric and trinuclear heterobimetallic MnIII···PtII···MnIII coordination compounds, prepared from the Ba[Pt(CN)4] and [Mn(L4A/B)(Cl)] (1a/b) precursor complexes, are reported. The polymeric complex [{Mn(L4A)}2{μ4-Pt(CN)4}]n (2a), where H2L4A = N,N’-ethylene-bis(salicylideneiminate), comprises the {Mn(L4A)} moieties covalently connected through the [Pt(CN)4]2− bridges, thus forming a square-grid polymeric structure with the hexacoordinate MnIII atoms. The trinuclear complex [{Mn(L4B)}2{μ-Pt(CN)4}] (2b), where H2L4B = N,N’-benzene-bis(4-aminodiethylene-salicylideneiminate), consists of two [{Mn(L4B)} moieties, involving pentacoordinate MnIII atoms, bridged through the tetracyanidoplatinate (II) bridges to which they are coordinated in a trans fashion. Both complexes possess uniaxial type of magnetic anisotropy, with D (the axial parameter of zero-field splitting) = −3.7(1) in 2a and −2.2(1) cm−1 in 2b. Furthermore, the parameters of magnetic anisotropy 2a and 2b were also thoroughly studied by theoretical complete active space self-consistent field (CASSCF) methods, which revealed that the former is much more sensitive to the ligand field strength of the axial ligands.
31. Tetranuclear Lanthanide Complexes Containing a Hydrazone-type Ligand. Dysprosium [2 × 2] Gridlike Single-Molecule Magnet and Toroic
Alexey Gusev, Radovan Herchel, Ivan Nemec, Victor Shul’gin, Igor Eremenko, Kostantin Lyssenko, Wolfgang Linert, and Zdeněk Trávníček, Inorg. Chem., 2016, 55 (23), pp 12470–12476. Citations.
A multidentate hydrazone-type ligand (Z,Z)-
bis(1-(pyridin-2-yl)-1-amino-methylidene)oxalohydrazide
(H2L) was utilized in the synthesis of three new isomorphous
tetranuclear complexes of the general formula
[Ln4(HL)4(H2L)2(NO3)4](NO3)4·4CH3OH (Ln = GdIII, 1,
TbIII, 2, DyIII, 3) with the gridlike [2 × 2] topology. The analysis of the static magnetic data revealed weak anti-ferromagnetic interaction among lanthanide(III) atoms,whereas dynamic magnetic data led to the observation of the single-molecule magnet behavior in zero static magnetic field for the Dy4 compound 3 with Ueff = 42.6 K and τ0 = 1.50 ×10−5 s. The theoretical CASSCF calculations supported also the presence of the net toroidal magnetic moment, which classifies
compound 3 also as a single-molecule toroic.
30. Solvent-induced structural diversity in tetranuclear Ni (II) Schiff-base complexes: the first Ni4 single-molecule magnet with a defective dicubane-like topology
Radovan Herchel, Ivan Nemec, Marek Machata and Zdeněk Trávníček, Dalton Trans., 2016, 45 (46), 18622-18634 Citations.
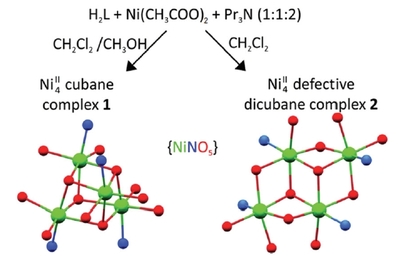
Two tetranuclear NiII complexes, namely [Ni4(L)4(CH3OH)3(H2O)]·CH3OH (1) and (Pr3NH)2[Ni4(L)4
(CH3COO)2] (2, Pr3N = tripropylamine), were synthesized from a tridentate Schiff base ligand H2L (2-[(E)-
(2-hydroxybenzylidene)amino]phenol) and Ni(CH3COO)2·4H2O, using different solvents and their ratios
(CH3OH and/or CH2Cl2). The prepared Ni4 complexes are of different structural types, involving an Ni4O4
cubane-like core (1) and Ni4O6 defective dicubane-like core (2), with all the Ni atoms hexacoordinated.
The complexes were characterized by elemental analysis, FT-IR spectroscopy, variable temperature and
field magnetic measurements, and single crystal X-ray analysis. The DFT and CASSCF/NEVPT2 theoretical
calculations were utilized to reveal information about the isotropic exchange parameters (Jij) and singleion
zero-field splitting parameters (Di, Ei). The variable temperature magnetic data suggested the competition
of the antiferromagnetic and ferromagnetic intracluster interactions in compound 1, which is in
contrast to compound 2, where all intracluster interactions are ferromagnetic resulting in the ground spin
state S = 4 with an easy-axis type of anisotropy quantified by the axial zero-field splitting parameter D =
−0.81 cm−1. This resulted in the observation of a field-induced slow-relaxation of magnetization (U =
3.3–6.7 K), which means that the complex 2 represents the first Ni4 single-molecule magnet with the
defective dicubane-like topology.
29. Ferromagnetic coupling mediated by Co⋯π non-covalent contacts in a pentacoordinate Co(II) compound showing field-induced slow relaxation of magnetization
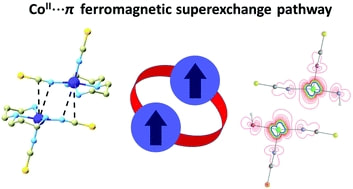
[CoII(dpt)(NCS)2], where dpt = bis(3-aminopropyl)amine, was identified as a pentacoordinate CoII compound showing field-induced slow relaxation of magnetization. Furthermore, intermolecular ferromagnetic coupling mediated by Co⋯π non-covalent contacts, where π orbitals originate from the thiocyanato ligand, is reported for the first time.
28. Magnetic anisotropy in pentacoordinate 2,6-bis(arylazanylidene-1-chloromethyl)pyridine cobalt(II) complexes with chlorido co-ligands
Ivan Nemec, Heng Liu, Radovan Herchel, Xuequan Zhang, Zdeněk Trávníček, Synth. Met. 2016, 215, 158–163. Citations.
27. Field-induced slow relaxation of magnetization in dinuclear and trinuclear CoIII•••MnIII complexes
Ivan Nemec, Radovan Herchel, Zdenek Travnicek and Tomas Silha , RSC Adv., 2016, 6, 3074-3083. Citations.
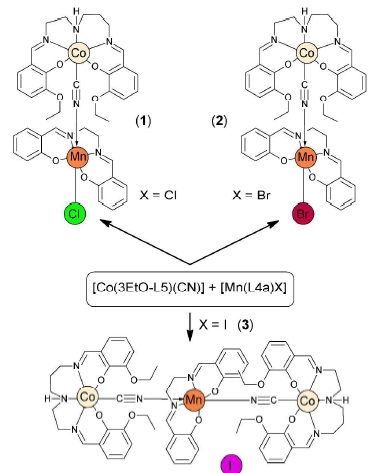
Two new dinuclear, [Co(3EtO-L5)(μ-CN)Mn(L4a)Cl] (1) and [Co(3EtO-L5)(μ-CN)Mn(L4a)Br] (2), and
two trinuclear [{Co(3EtO-L5)(μ-CN)}2{Mn(L4a)]I (3) and [{Co(3EtO-L5)(μ-CN)}2Mn(L4a)](NO3) (4)
complexes were prepared and thoroughly characterized (H23EtO-L5 = N,N′-bis(3-ethoxy-2-
hydroxybenzylidene)-1,6-diamino-3-azahexane), L4a2- = N,N′-ethane-bis(salicylideneiminate) dianion).
The crystal structures were determined for all four compounds, while the static and dynamic magnetic
properties were studied only for compounds 1–3. It has been revealed by simultaneous fitting of
temperature and field dependent magnetic data and by using the spin Hamiltonian formalism involving
the axial anisotropy term that the manganese(III) atoms possess relatively large and negative axial
magnetic anisotropy in 1–3, with D = −3.9(2) cm-1 in 1, −4.9(2) cm-1 in 2, and −4.1(1) cm-1 in 3. These
results were supported by ab initio CASSCF calculations which were in a good agreement with the
experimental ones, however, a small rhombicity was calculated contrary to the experimental evaluations:
Dcalc (Ecalc/Dcalc) = −3.2 cm-1 (0.04) in 1, −3.0 cm-1 (0.03) in 2, and −3.6 cm-1 (0.04) in 3. The
measurements of dynamic magnetic data confirmed that compounds 1–3 represent a new type of Mn(III) field-induced single-ion magnets. The peak maxima of the frequency dependent out-phase susceptibility
were below the lowest accessible temperature in all the three cases (i.e. below 1.9 K) and this prevented
us from the construction of the Argand diagram. Nevertheless, the approximate procedure for extracting
the spin-reversal barrier (Ueff) and relaxation time (τ0) was used, and the values of Ueff ranging from 11 to 20 K and t0 from 0.1 to 19 x 10-7 s were obtained.
2015
26. Experimental and Theoretical Investigations of Magnetic Exchange Pathways in Structurally Diverse Iron(III) Schiff-Base Complexes
Radovan Herchel, Ivan Nemec, Marek Machata and Zdenek Travnicek, Inorg. Chem., 2015, 54 (17), pp 8625–8638. Citations.
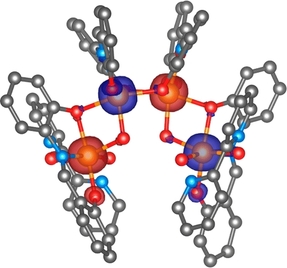
Polynuclear iron(III) complexes were prepared by utilizing mononuclear precursors containing a Schiff-base ligand (2-hydroxyphenylsalicylaldimine) and 2,2′-bipyridine-N-dioxide. Investigations of their X-ray structures, followed by DFT calculations of magnetic exchange parameters, revealed superexchange pathways going through intramolecular/intermolecular noncovalent interactions (π−π stacking, C−H···O contacts and O−H···O hydrogen bonds, diamagnetic metal cation) and/or covalent bonds {(μ-OPh, μ-OH) or bis(μ-OPh) bridging modes}, which helped us to postulate the spin Hamiltonians for magnetic analysis of experimental data in a trustworthy manner.
25. Field-induced slow relaxation of magnetization in pentacoordinate Co(II) compound [Co(phen)(DMSO)Cl2]
Ivan Nemec, Raphael Marx, Radovan Herchel, Petr Neugebauer, Joris van Slageren, Zdenek Travnicek , Dalton Trans. 2015, 44 (33), 15014-15021. Citations.
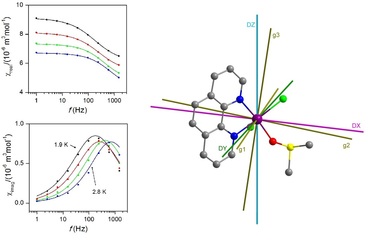
The static and dynamic magnetic properties of the pentacoordinate [Co(phen)(DMSO)Cl2] compound
(phen = 1,10’- phenantroline, DMSO = dimethyl sulfoxide) were thoroughly studied by experimental
(SQUID magnetometry and HF-EPR spectroscopy) and theoretical methods (DFT and CASSCF
10 calculations). It has been found from temperature/field-dependent magnetization measurements that the
studied compound possesses a large and negative magnetic anisotropy (D = -17(1) cm-1) with large
rhombicity (E/D = 0.24(5)), and these experimental results are in agreement with ab initio calculations (D
= -17.7 cm-1, E/D = 0.31). Interdoublet resonances were not observed in the HF-EPR measurements, but
the large rhombicity was confirmed (DEPR = -17.7 cm-1 (fixed from CASSCF calculation), E/DEPR = 0.33).
15 A frequency dependent out-of-phase susceptibility signal was observed only in non-zero static magnetic
field (B = 0.1 T) and the following parameters of slow-relaxation of magnetisation were derived from the
experimental data: either the energy of spin reversal barrier, Ueff = 10.4 K, and the relaxation time, τ0 =
5.69×10-9 s using Debye model, or Ueff = 21.4-40.3 K and τ0 = 0.248-58.3×10-9 based on a simplified
model.
24. Suppressing of slow magnetic relaxation in tetracoordinate Co(II) field-induced single-molecule magnet in hybrid material with ferromagnetic barium ferrite
I. Nemec, R. Herchel, Z. Travnicek, Sci. Rep., 2015, 5, 10761. Citations.
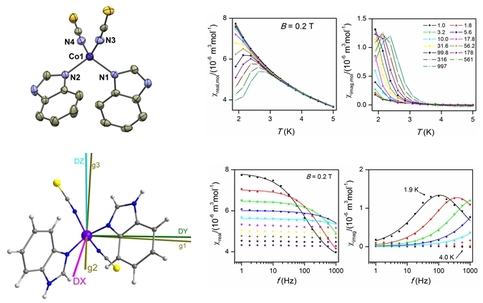
The novel field-induced single-molecule magnet based on a tetracoordinate mononuclear heteroleptic Co(II) complex involving two heterocyclic benzimidazole (bzi) and two thiocyanido ligands, [Co(bzi)2(NSC)2], (CoL4), was prepared and thoroughly characterized. The analysis of AC susceptibility data resulted in the spin reversal energy barrier U = 14.7 cm−1, which is in good agreement with theoretical prediction, Utheor. = 20.2 cm−1, based on axial zero-field splitting parameter D = −10.1 cm−1 fitted from DC magnetic data. Furthermore, mutual interactions between CoL4 and ferromagnetic barium ferrite BaFe12O19 (BaFeO) in hybrid materials resulted in suppressing of slow relaxation of magnetization in CoL4 for 1:2, 1:1 and 2:1 mass ratios of CoL4 and BaFeO despite the lack of strong magnetic interactions between two magnetic phases.
23. Large and negative magnetic anisotropy in pentacoordinate mononuclear Ni(II) Schiff base complexes
Ivan Nemec, Radovan Herchel, Ingrid Svoboda, Roman Boca, Zdenek Travnicek, Dalton Trans. 2015, 44, 9551-9560. Citations.
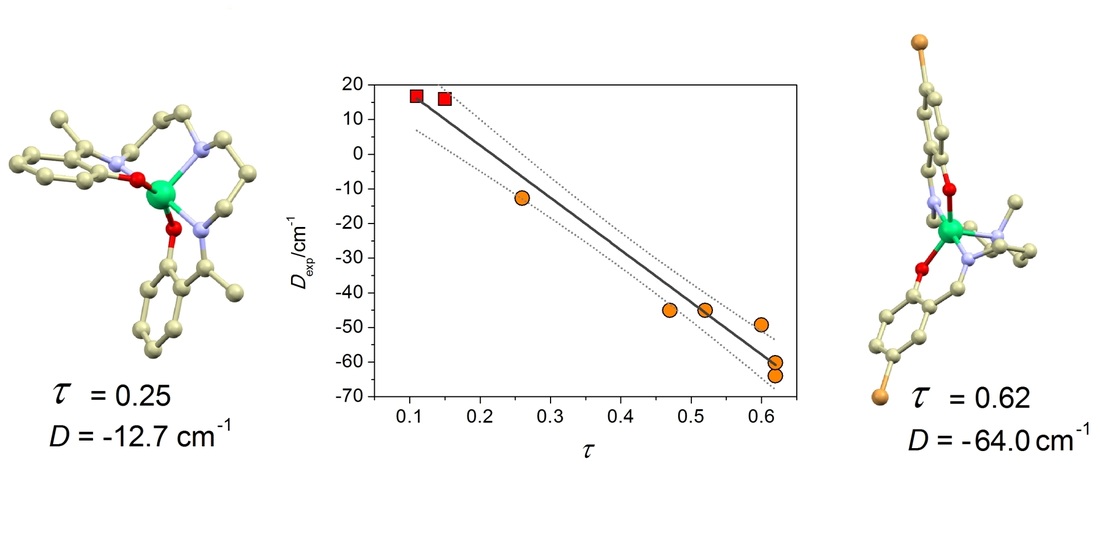
A series of pentacoordinate Ni(II) complexes of the general formula [Ni(L5)] (1–6) with various pentadentate Schiff base ligands H2L5 (originating in a condensation of aromatic ortho-hydroxy-aldehydes and aliphatic triamines) was synthesized and characterized by X-ray structure analysis and magnetometry. The alternations of substituents on the H2L parent ligand resulted in the complexes with the geometry varying between the square-pyramid and trigonal-bipyramid. In the compounds whose chromophore geometry is closer to a trigonal-bipyramid, a large and negative uniaxial anisotropy (D = −64 cm−1) was identified. Moreover, the simple linear expression for the axial zero-field splitting (ZFS) parameter, D/cm−1 = 32.7(4.8) − 151(10)τ, was proposed, where τ (in degrees) stands for the Addison parameter. The results of magnetic analysis were also supported by ab initio CASSCF/NEVPT2 calculations of the ZFS splitting parameters D and E, and g tensors. Despite large and negative D-values of the reported compounds, slow relaxation of magnetization was not observed either in zero or non-zero static magnetic field, thus no single-molecule magnetic behaviour was detected.
22. Relationship between strength of hydrogen bonding and spin crossover behaviour in a series of iron(III) Schiff base complexes
Ivan Nemec, Radovan Herchel and Zdeněk Trávníček, Dalton Trans. 2015, 44, 4474-4484. Citations.
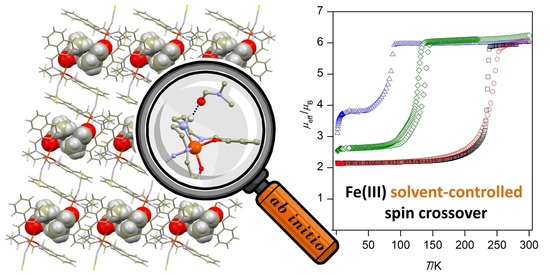
X-ray crystal structures and magnetic properties of an isostructural series of iron(III) Schiff base complexes with the general formula [Fe(L5)(NCR)]•Solv (where H2L5 = N,N'-bis(2-hydroxy-naphthylidene)-1,6-diamino-4-azahexane, R = S, Solv = tetrahydrofuran, 1a; R = S, Solv = methanol and 0.5 pyrazine, 1b; R = S, Solv = butanone, 1c; Solv = N,N’-dimethylformamide, R = S (1d) or R = Se (1d’); R = S, Solv = dimethyl sulfoxide, 1e) are reported. In the crystals, the individual [Fe(L5)(NCR)] molecules are connected through weak C-H•••O, C–H•••π or C-H•••S non-covalent contacts into 2D supramolecular networks, while the guest-solvent (Solv) molecules are trapped into the cavities between two adjacent layers, which are furthermore stabilized by N–H•••O hydrogen bonds connecting the Solv oxygen atom with the amine group of the [Fe(L5)(NCR)] molecule, with the N•••O distances varying from 2.921(6) Å (in 1d’) to 3.295(2) Å (in 1a). The magnetic properties of the complexes were tuned by the different Solv molecules and as a result of this, four new spin crossover (SCO) compounds with cooperative spin transitions are reported, which are accompanied by thermal hysteresis in two cases (1d and 1e): 1c, T1/2 = 84 K, 1d, T1/2↓ = 232 K, T1/2↑ = 235 K and 1e, T1/2↓ = 127 K T1/2↑ = 138 K. The role of the N–H•••O hydrogen bonding in the occurrence and tuning of SCO was also computationaly studied using a topological analysis, and also by evaluation of non-covalent interaction (NCI) indexes. Both theoretical approaches showed clear relationship between the strength of the N–H•••O hydrogen bonds and T1/2, as already followed from X-ray structural and magnetic data.
21. Iron(III) Complexes with Pentadentate Schiff-base Ligands: Influence of Crystal Packing Change and Pseudohalido Coligand Variations on Spin Crossover
Christoph Krüger, Peter Augustín, Ľubor Dlháň, Ján Pavlik, Ján Moncoľ, Ivan Nemec, Roman Boča and Franz Renz, Polyhedron, 2015, pp. 194-20. Citations.
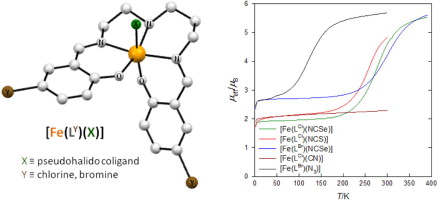
Two novel iron(III) complexes involving pentadentate Schiff-base ligands, [Fe(LBr)(L1)], show temperature induced incomplete spin crossover of a gradual nature. While for the L1 = NCSe− coligand the transition temperature (326 K or 317 K) lies above room temperature, the N3− coligand causes its drop down to 143 K or 140 K (two values were obtained by different models). This shift is associated with a significant decrease of the enthalpy of the transition from about ΔH = 6.1 kJ mol−1 to ca. ΔH = 1.7 kJ mol−1, while the entropy of the transition is about ΔS = 19 J K−1 mol−1 and ΔS = 12 J K−1 mol−1, respectively. Two analogous complexes with Cl− and NCS− coligands remain high-spin over the whole temperature range.
20. Spin Crossover and High Spin Electroneutral Mononuclear Iron(III) Schiff Base Complexes involving Terminal Pseudohalido Ligands
Petra Masárová, Pavel Zoufalý, Jan Moncoľ, Ivan Nemec, Jan Pavlik, Milan Gembický, Zdeněk Trávníček, Roman Boča, Ivan Šalitroš, New J. Chem, 2015, 39, 508-519. Citations.
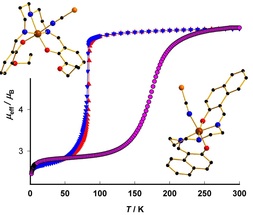
Investigations on a series of six novel mononuclear iron(III) Schiff base complexes with the general formula [Fe(L)X] (where H2L
is a pentadentate Schiff-base ligand, X = pseudohalido ligand) are reported. Several different aromatic 2-hydroxyaldehyde
10 derivatives were used in combination with N,N’−bis(2−aminoethyl)−1,3−propanediamine (compounds 1−5) and
2,2'−diaminodiethylamine (for 6) to synthesize the H2L Schiff base ligands. The consecutive reactions with iron(III) chloride
resulted in the preparation of the [Fe(L)Cl] precursor complexes which were left to react with pseudohalido ligands (NCS−
for 1,
2, 3, 4, 6; N3
−
for 4). Structural investigations revealed an usual coordination of the pentadentate Schiff base ligands via N3O2
donor atoms and sixth coordination place is occupied by the N donor of the corresponding pseudohalido ligand. The spin
15 crossover was observed in two cases with transition temperatures: Tc = 83 K (hysteresis width of ∆T = 2 K) for 1 and Tc = 174 K
for 2. Magnetic investigations of compounds 3−6 revealed high spin behaviour. The magnetic data of all compounds were
analysed using the spin Hamiltonian formalism including the zero-field splitting term and molecular field effect. In the case of the
spin crossover complexes 1 and 2, the Ising-like model was applied
2014
19. Novel Schiff Bases Based on the Quinolinone Skeleton: Syntheses, X-ray Structures and Fluorescent Properties
Zdeněk Trávníček , Roman Buchtík and Ivan Nemec, Molecules, 2014, 19(9), 13509-13525. Citations.
A series of a new type of Schiff bases 1–7, derived from 2-phenyl-3-amino-4(1H)-quinolinone and R-salicyladehyde derivatives wherein R = 3-hydroxy (1), 3,4-dihydroxy (2), 3-methoxy (3), 3-carboxy (4), 3-allyl (5), 5-chloro (6), and 5-nitro (7), was synthesized and structurally characterized. Each of the molecules 1, 3 and 7 consists of three planar moieties (i.e., a quinolinone and two phenyl rings), which are mutually oriented differently depending on the appropriate substituent R and the extent of non-covalent contacts stabilizing the crystal structures. The compounds were studied for their fluorescence properties, where compound 6 yielded the strongest intensity both in the solid phase and in 100 μM ethanol solution with a quantum yield of φ = 3.6% as compared to quinine sulfate used as a standard. The in vitro cytotoxicity of these compounds was tested against the human osteosarcoma (HOS) and breast adenocarcinoma (MCF7) cell lines, revealing no activity up to the concentration of 50 µM.
18. Towards better understanding of magnetic exchange mediated by hydrogen bonds in Mn(III)/Fe(III) salen-type supramolecular dimers
Ivan Nemec Radovan Herchel Tomáš Šilha and Zdeněk Trávníček, Dalton Trans. 2014, 43, 15602-15616. Citations.
The thorough study of structural and magnetic properties were performed on a series of trinuclear and
dinuclear M(III)/Fe(III) complexes consisting of [M(L4)(Solv)]+ and [Fe(CN)5(NO)]2- moieties (M =
Fe(III) or Mn(III), Solv = H2O or CH3OH, L4 = tetradentate salen-type ligands), in which dominant
magnetic exchange is mediated by OS–H···OPh hydrogen bonds in [M(L4)(Solv)]+···[M(L4)(Solv)]+
10 supramolecular dimers. As deduced from magnetic analysis involving also determination of zero-field
splitting (ZFS) parameters for Mn(III) and Fe(III) ion as well as from comprehensive DFT calculations
and modelling, it may be concluded that the strength of magnetic exchange is correlated with a number of
hydrogen bonds and with the OPh⋅⋅⋅OS distance between the phenolic oxygen of salen-type ligand (OPh)
and oxygen of the solvent coordinated to the adjacent metal atom (OS)
dinuclear M(III)/Fe(III) complexes consisting of [M(L4)(Solv)]+ and [Fe(CN)5(NO)]2- moieties (M =
Fe(III) or Mn(III), Solv = H2O or CH3OH, L4 = tetradentate salen-type ligands), in which dominant
magnetic exchange is mediated by OS–H···OPh hydrogen bonds in [M(L4)(Solv)]+···[M(L4)(Solv)]+
10 supramolecular dimers. As deduced from magnetic analysis involving also determination of zero-field
splitting (ZFS) parameters for Mn(III) and Fe(III) ion as well as from comprehensive DFT calculations
and modelling, it may be concluded that the strength of magnetic exchange is correlated with a number of
hydrogen bonds and with the OPh⋅⋅⋅OS distance between the phenolic oxygen of salen-type ligand (OPh)
and oxygen of the solvent coordinated to the adjacent metal atom (OS)
17. Versatile coordination modes of bis[5-(2-pyridine-2-yl)-1,2,4-triazole-3-yl]alkanes in Cu(II) complexes
Alexey N. Gusev, Ivan Nemec, Radovan Herchel, Eziz Bayjyyev, Galyna N. Nyshchimenko, Grigory G. Aleksandrov, Igor Leonidovich Eremenko, Zdenek Trávníček, Miki Hasegawa and Wolfgang Linert ; Dalton. Trans., 2014, 43, 7153-7165. Citations.
Nine new mononuclear and polynuclear Cu(II) complexes containing [5-(2-pyridine-2-yl)-1,2,4-triazole-3-yl]alkanes (H2Ln, n = 1–4) in combination with other ligands, were synthesized and characterized by various techniques such as elemental analysis, FTIR, NMR and UV-Vis spectroscopy. X-ray structures of H2L3 and H2L4 as well as complexes 1–8 were determined. The X-ray structures revealed that relatively small composition and structural changes in the H2Ln ligands have a substantial impact on the coordination geometries of the complexes themselves as well as on their resulting magnetic properties.
16. A zinc(II) quinolinone complex (Et3NH)[Zn(qui)Cl2]: Synthesis, X-ray structure, spectral properties and in vitro cytotoxicity
Roman Buchtík, Ivan Nemec, Zdeněk Trávníček; J. Mol. Struct. 2014; 1060:42-48. Citations.
Roman Buchtík, Ivan Nemec, Zdeněk Trávníček; J. Mol. Struct. 2014; 1060:42-48. Citations.
2013
15. Investigation of magnetic exchange pathways in heterotrinuclear manganese(III) Schiff base complexes involving tetrathiocyanidoplatinate(II) bridges
Nemec I., Šilha T., Herchel R. and Trávníček Z.; Eur. J. Inorg. Chem.; 2013, (33), 5781-5789. Citations.
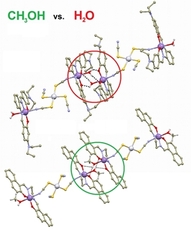
Three new trinuclear manganese(III) Schiff base complexes with the tetrathiocyanidoplatinum(II) bridges were prepared. The coordinated solvent ligands form intermolecular hydrogen bonds resulting into 1D chain structures. The antiferromagnetic exchange interactions within the chains mediated by H-bonds were found to be stronger for the compounds with the water ligands (1c) than for those involving the methanol molecules (1a-b), as determined by interpretations of magnetic data and DFT calculations.
14. Cobalt(III) Schiff-base cyanido complex usable as a ligand in preparation of heterobimetallic Co(III)-Fe(III) building blocks
Nemec, I.; Zoufalý, P. ; Herchel R. and Trávníček Z.; Inorg. Chem. Commun. 2013, 35, 50-53, Citations.
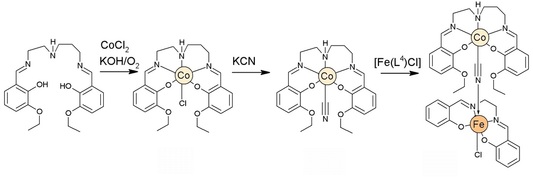
X-ray structure and magnetic properties of a novel dinuclear heterobimetallic CoIII–CN–FeIII compound of the composition [{Co(3EtO-L5)}(μ-CN){Fe(L4)(Cl)}] (2) is reported. Adjacent complex molecules are connected through intermolecular N–H⋅⋅⋅Cl hydrogen bonds into 1D supramolecular chains. Such a compound can be considered as a potential building block for the preparation of Prussian blue analogues of high nuclearity showing interesting magnetic properties.
13. Structural and magnetic characterizations of the first manganese(III) Schiff base complexes involving hexathiocyanidoplatinate(IV) bridges
Šilha, T.; Nemec, I.; Herchel, R.; Trávníček, Z.; CrystEngComm. 2013, 15, 5351-5358, Citations.
12. Spin crossover in iron(III) complexes with pentadentate Schiff-base ligands and pseudhalido coligands
Krüger, C.; Augustín, P.; Nemec, I.; Trávníček, Z.; Oshio, H.; Boča, R.; Renz, F., Eur. J. Inorg. Chem. 2013, Issue: 5-6 Pages: 902-915, Citations.
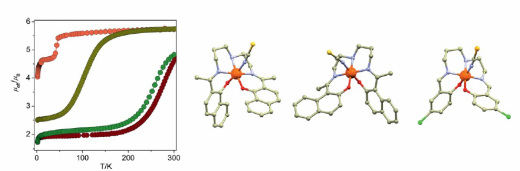
Iron(III) complexes that involve pentadent- a magnetochemical investigation. Four
ate Schiff base ligands and chlorido, azido, complexes with the thiocyanato or selenocyanido,
cyanato, thiocyanato, or selenocy- cyanato ligand exhibit the spin crossover,
746 anato coligands have been synthesized, centered at the critical temperature (Tc) of
structurally characterized, and subjected to 42, 114, 282, and 293 K, respectively.
2012
11. Mixed-valence heptanuclear iron complexes with ferromagnetic interaction
Šalitroš, I.; Boča, R.; Herchel, R.; Moncol, J.; Nemec, I.; Ruben, M.; Renz, F., Inorg. Chem., 2012, Volume: 51 Issue: 23 Pages: 12755-12767, Citations.
10. A new family of Fe2Ln complexes built from mononuclear anionic Schiff base subunits
Nemec, I.; Machata, M.; Herchel, R.; Boca, R.; Travnicek, Z., Dalton Trans. 2012, 41 (48), 1460314 610. Citations
9. Anion driven modulation of magnetic intermolecular interactions and spin crossover properties in an isomorphous series of mononuclear iron(iii) complexes with a hexadentate Schiff base ligand
Nemec, I.; Herchel, R.; Šalitroš, I.; Trávníček, Z.; Moncoľ, J.; Fuess, H.; Ruben, M.; Linert, W., CrystEngComm 2012, 14 (20), 7015-7024. Citations.
8. Direct determination of zero-field splitting in Co(II) complexes by FAR infrared spectroscopy.
Šebová, M.; Boča, R.; Dlháň, Ľ.; Nemec, I.; Papánková, B.; Pavlik, J.; Fuess, H., Inorg. Chim. Acta 2012, 383 (0), 143-151. Citations.
Three centrosymmetric, mononuclear cobalt(II) complexes possessing large zero-field splitting parameter D have been studied by means temperature-dependent FAR infrared spectroscopy. From 15 temperature-dependent absorption peaks referring to the transition between the six lowest crystal-field multiplets the value of the D-parameter has been directly read off. These data are comparable with those values extracted by fitting the magnetic susceptibility and magnetization data.
2011
7. Tuning of spin crossover behaviour in iron(iii) complexes involving pentadentate Schiff bases and pseudohalides
Nemec, I.; Herchel, R.; Boča, R.; Travníček, Z.; Svoboda, I.; Fuess, H.; Linert, W., Dalton Transactions 2011, 40 (39), 10090-10099. Citations.
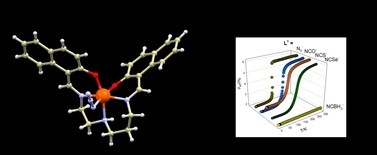
A series of mononuclear iron(III) complexes with pentadentate Schiff base ligand and pseudohalido co-ligand was prepared and characterized. For the first time tuning of the spin crossover phenomenon by pseudohalido ligand exchange was performed on iron(III) complexes. Azido compound exhibits cooperative spin transition with narrow hysteresis.
6. Heterobimetallic assemblies of Ni(II) complexes with a tetradentate amine ligand and diamagnetic cyanidometallates
Nemec, I.; Herchel, R.; Boča, R.; Svoboda, I.; Travníček, Z.; Dlháň, L.; Matelková, K.; Fuess, H., Inorg. Chim. Acta 2011, 366 (1), 366-372. Citations.
2010
5. Magneto-structural relationships for a mononuclear Co(II) complex with large zero-field splitting
Papánková, B.; Boča, R.; Dlháň, L.; Nemec, I.; Titiš, J.; Svoboda, I.; Fuess, H., Inorg. Chim. Acta 2010, 363 (1), 147-156. Citations.
2009
4. High-spin Schiff-base dinuclear iron(III) complexes bridged by N-oxide ligands
Nemec, I.; Boča, R.; Gembický, M.; Dlháň, L.; Herchel, R.; Renz, F., . Inorg. Chim. Acta 2009, 362 (13), 4754-4759. Citations.
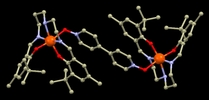
A series of dinuclear Fe(III) complexes bridged by N-oxide heterocyclic linkers was prepared and characterized. Magnetic behaviour corresponds to the high spin dimeric molecule with weak intradimer antiferromagnetic interaction and S = 5 ground state. The strength of the exchange interaction, as can be expected, depends from the length of the bridging ligand.
3. Interplay between spin crossover and exchange interaction in iron(lll) complexes
Boča, R.; Nemec, I.; Šalitroš, I.; Pavlik, J.; Herchel, R.; Renz, F., Pure and Applied Chemistry 2009, 81 (8), 1357-1383. Citations.
2. Unconventional spin crossover in dinuclear and trinuclear iron(III) complexes with cyanido and metallacyanido bridges
Šalitroš, I.; Boča, R.; Dlháň, L.; Gembický, M.; Kožíšek, J.; Linares, J.; Moncol, J.; Nemec, I.; Perašínová, L.; Renz, F.; Svoboda, I.; Fuess, H., Eur. J. Inorg. Chem. 2009, (21), 3141-3154. Citations.
1. Dinuclear Fe(III) complexes with spin crossover
Nemec, I.; Boča, R.; Herchel, R.; Travníček, Z.; Gembický, M.; Linert, W., Mon. Chem. 2009, 140 (7), 815-828. Citations.
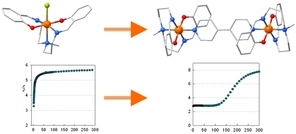
Upon reaction of mononuclear chlorido precursor complexes with bridging organic ligands, e.g. 4,4'-bipyridyl, the dinuclear precursor complexes were prepared. Effect of the bridging ligand rigidity manifested itself in complete and more abrupt spin transitions observed for compounds with conjugated bridging ligands, while the compounds with non-conjugated bridges have incomplete and very gradual transitions.